Stable And Unstable Nuclei Radioactivity Physics Fuseschool

Stable Unstable Nuclei Ms Pdf Isotope Radioactive Decay In this video we are going to learn about radioactive decay and that unstable nuclei either have too many protons or too many neutrons, which upsets the strong nuclear forces holding the atom. But not all nuclei are stable. unstable nuclei either have too many protons or too many neutrons, upsetting the strong nuclear forces. unstable nuclei try to balance themselves by giving off the excess proton or neutron. this is radioactive decay. unstable nuclei are radioactive and emit radiation.

Stable And Unstable Nuclei Radioactivity Physics Fuseschool On Join thousands of students who trust us to help them ace their exams! watch the first video. Explore why some nuclei are stable while others undergo radioactive decay. learn how neutron to proton ratio and nuclear forces determine nuclear stability. The unstable nuclei, in order to become stable nuclei, emit particles and or electromagnetic radiation. these types of nuclei are said to be radioactive, and the emission is called radioactivity. Many nuclei are stable. many are radioactive or unstable, and will tend to decay by emitting a particle, transforming the atom (made up of protons, neutrons and electrons1) into an atom of another element, or into a lower energy state.
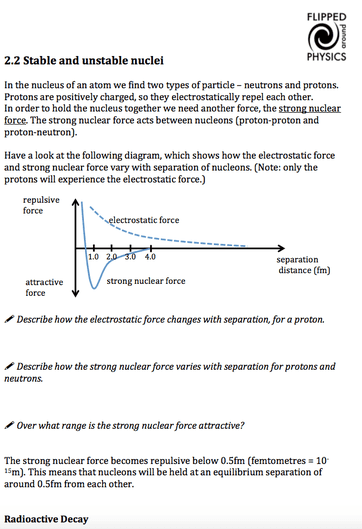
Stable And Unstable Nuclei A Level Physics Flipped Around Physics The unstable nuclei, in order to become stable nuclei, emit particles and or electromagnetic radiation. these types of nuclei are said to be radioactive, and the emission is called radioactivity. Many nuclei are stable. many are radioactive or unstable, and will tend to decay by emitting a particle, transforming the atom (made up of protons, neutrons and electrons1) into an atom of another element, or into a lower energy state. The unstable nuclei lie above and below the line of stability in the neutron – proton plot. this gives information of the type of radioactive decay they will undergo. nuclei which lie above the line of stability contain too many neutrons to be stable. they are referred to as “neutron rich”. However, just when the first direct evidence of atoms was obtained, it became clear that they have a substructure and a tiny nucleus. the nucleus itself has spectacular characteristics. for example, certain nuclei are unstable, and their decay emits radiations with energies millions of times greater than atomic energies. 2.2 stable and unstable nuclei in the nucleus of an atom, we find two types of particle – neutrons and protons. protons are positively charged, so they electrostatically repel each other. in order to hold the nucleus together we need another force, the strong nuclear force. In a nuclear decay reaction, also called radioactive decay, an unstable nucleus emits radiation and is transformed into the nucleus of one or more other elements. the resulting daughter nuclei have a lower mass and are lower in energy (more stable) than the parent nucleus that decayed.

Stable And Unstable Nuclei A Level Physics Flipped Around Physics The unstable nuclei lie above and below the line of stability in the neutron – proton plot. this gives information of the type of radioactive decay they will undergo. nuclei which lie above the line of stability contain too many neutrons to be stable. they are referred to as “neutron rich”. However, just when the first direct evidence of atoms was obtained, it became clear that they have a substructure and a tiny nucleus. the nucleus itself has spectacular characteristics. for example, certain nuclei are unstable, and their decay emits radiations with energies millions of times greater than atomic energies. 2.2 stable and unstable nuclei in the nucleus of an atom, we find two types of particle – neutrons and protons. protons are positively charged, so they electrostatically repel each other. in order to hold the nucleus together we need another force, the strong nuclear force. In a nuclear decay reaction, also called radioactive decay, an unstable nucleus emits radiation and is transformed into the nucleus of one or more other elements. the resulting daughter nuclei have a lower mass and are lower in energy (more stable) than the parent nucleus that decayed.

Stable And Unstable Nuclei A Level Physics Flipped Around Physics 2.2 stable and unstable nuclei in the nucleus of an atom, we find two types of particle – neutrons and protons. protons are positively charged, so they electrostatically repel each other. in order to hold the nucleus together we need another force, the strong nuclear force. In a nuclear decay reaction, also called radioactive decay, an unstable nucleus emits radiation and is transformed into the nucleus of one or more other elements. the resulting daughter nuclei have a lower mass and are lower in energy (more stable) than the parent nucleus that decayed.
Comments are closed.