Specific Heat Lab Calculations

Specific Heat Lab Pdf In this experiment, students will find the specific heat of three “unknown” samples of metal. to do this, students will use their knowledge of thermodynamics to construct a calorimeter. The specific heat of a substance can be used to calculate the temperature change of the substance when it is heated or cooled. specific heat calculations are illustrated.
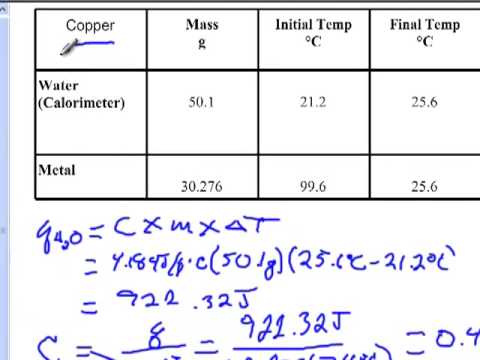
Specific Heat Lab In this experiment, you will determine the specific heat of a metal sample. the metal sample will be heated to a high temperature then placed into a calorimeter containing a known quantity of water at a lower temperature. The relationship between the heat supplied (q) to the mass (m), the temperature increase (dt) and the specific heat (c) of the substance is given by the following equation. Lab manual for determining the specific heat of a metal. includes procedure, data tables, calculations, and follow up questions. high school early college level. We can calculate the specific heat of the unknown metal (cm) using the following equation. since we are solving for cm and not the final temperature, we can use absolute value for the left side of the equation.

Specific Heat Capacity Worksheet Worksheet Worksheets Library Specific heat is the amount of energy required to increase the temperature of 1 gram of a substance by 1 oc (molar heat capacity is similar, but for 1 mole of a substance). different substances require different amounts of energy to increase temperature, as shown in table 1 below. Measuring the initial and final temperatures of the systems, and knowing the specific heat capacities of the cup, stirrer, and water, you will be able to calculate the specific heat capacity of the metal shot. The specific heat of a substance can be used to calculate the temperature change that a given substance will undergo when it is either heated or cooled. the equation that relates heat (q) to specific heat (c p), mass (m), and temperature change (Δ t) is shown below. The amount of heat absorbed or released (q) by the object depends on its mass (m), specific heat (cs), and the change in temperature (Δt). specific heat can be defined as the amount of heat required (q) to raise the temperature of one gram of the substance by one degree celsius.
Comments are closed.