Solved The Liquid Water Is Kept At 25%d1%82%d0%b8%d1%88c A What Is The Vapor Chegg

Solved The Liquid Water Is Kept At 25тишc A What Is The Vapor Chegg Question: the liquid water is kept at 25∘c a) what is the vapor pressure according to the table? (5 pts) b) what is the vapor pressure according to antoine equation? (10 pts) c) which of those two data is more accurate?. For water at 25 °c, the vapor pressure of 0.0313 atm indicates the tendency of water molecules to escape from the liquid phase into the gas phase. this concept is crucial for understanding the behavior of substances in equilibrium and is foundational for calculating equilibrium constants.

Solved The Vapor Pressure Of Water At 25 Degree C Is 3160 Chegg The only source of pressure is the vapor in equilibrium with water. at 25 o c the vapor pressure of water is 3.17 kpa and thus the pressure in the vessel is 3.17 kpa. Determine the enthalpy and entropy changes of liquid water for a change of state from 1 bar and 25 ºc to 1000 bar and 50 ºc. the following data for water are available:. A 2 m 3 tank initially contains a mixture of saturated vapor steam and saturated liquid water at 3000 kpa. of the total mass, 10 % is vapor. saturated liquid water is bled from the tank through a valve until the total mass in the tank is 40 % of the initial total mass. Some of the water that you drink may eventually be converted into sweat and evaporate. if you drink a 20 ounce bottle of water that had been in the refrigerator at 3.8 °c, how much heat is needed to convert all of that water into sweat and then to vapor??.

Solved 25pts ï At 25â C ï Liquid Water Is In Equilibrium Chegg A 2 m 3 tank initially contains a mixture of saturated vapor steam and saturated liquid water at 3000 kpa. of the total mass, 10 % is vapor. saturated liquid water is bled from the tank through a valve until the total mass in the tank is 40 % of the initial total mass. Some of the water that you drink may eventually be converted into sweat and evaporate. if you drink a 20 ounce bottle of water that had been in the refrigerator at 3.8 °c, how much heat is needed to convert all of that water into sweat and then to vapor??. A closed vessel contains pure water, in thermal equilibrium with its vapour at 25°c (stage #1), as shown. the vessel in this stage is then kept inside an isothermal oven which is having an atmosphere of hot air maintained at 80°c. Raoult’s law, formulated by french chemist françois marie raoult in 1887, describes how the vapor pressure of an ideal solution is proportional to the mole fraction of the solvent. Even more energy is required to vaporize water; it would take 2256 kj to change 1 kg of liquid water at the normal boiling point (100º c 100º c at atmospheric pressure) to steam (water vapor). Using a level meter (a device with an air bubble between two marks of a horizontal water tube) it can shown that the road that looks uphill to the eye is actually downhill. 1 4c there is no truth to his claim. it violates the second law of thermodynamics.
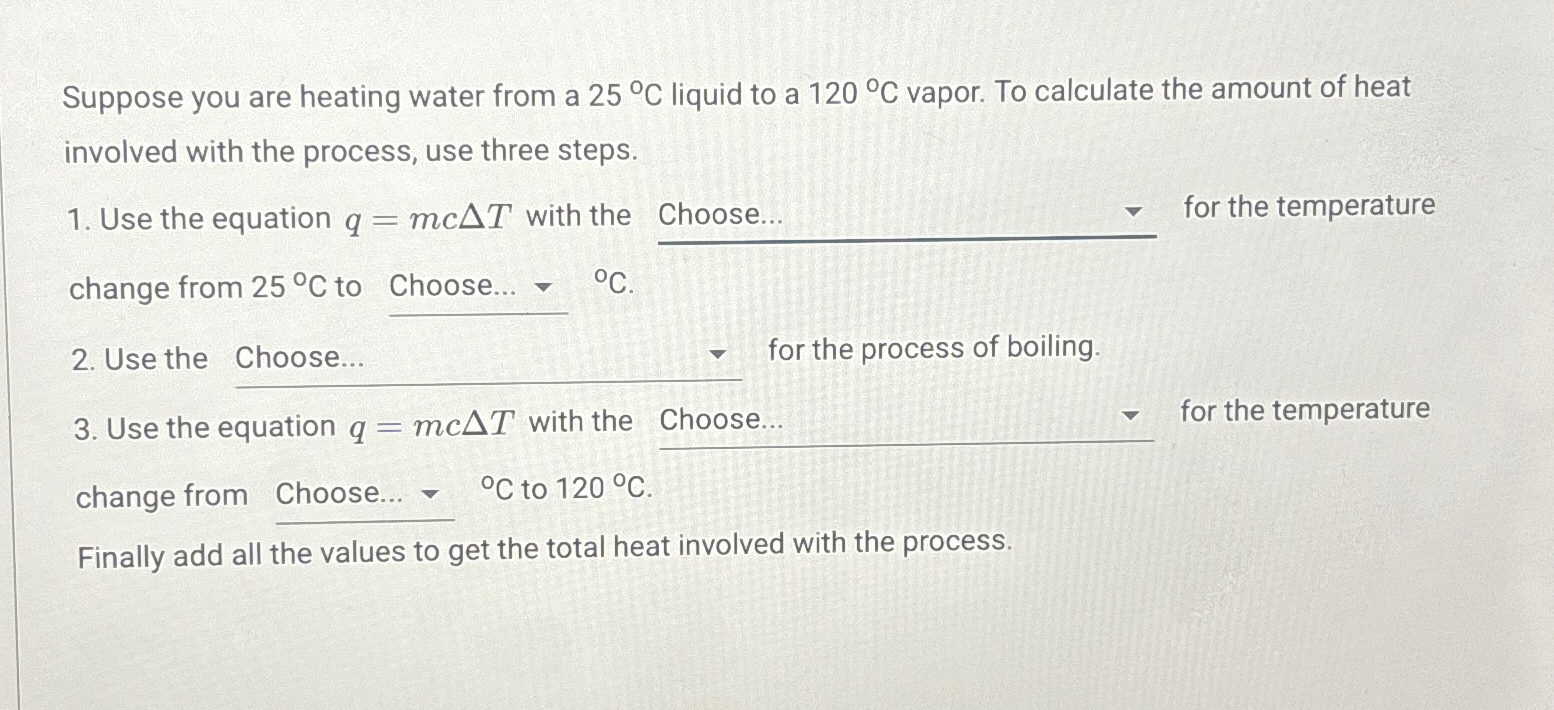
Solved Suppose You Are Heating Water From A 25â C ï Liquid To Chegg A closed vessel contains pure water, in thermal equilibrium with its vapour at 25°c (stage #1), as shown. the vessel in this stage is then kept inside an isothermal oven which is having an atmosphere of hot air maintained at 80°c. Raoult’s law, formulated by french chemist françois marie raoult in 1887, describes how the vapor pressure of an ideal solution is proportional to the mole fraction of the solvent. Even more energy is required to vaporize water; it would take 2256 kj to change 1 kg of liquid water at the normal boiling point (100º c 100º c at atmospheric pressure) to steam (water vapor). Using a level meter (a device with an air bubble between two marks of a horizontal water tube) it can shown that the road that looks uphill to the eye is actually downhill. 1 4c there is no truth to his claim. it violates the second law of thermodynamics.

Solved The Vapor Pressure Of Water At 25 C Is 0 0313 Atm Chegg Even more energy is required to vaporize water; it would take 2256 kj to change 1 kg of liquid water at the normal boiling point (100º c 100º c at atmospheric pressure) to steam (water vapor). Using a level meter (a device with an air bubble between two marks of a horizontal water tube) it can shown that the road that looks uphill to the eye is actually downhill. 1 4c there is no truth to his claim. it violates the second law of thermodynamics.

Solved Thermodynamics 18 Pts A The Vapor Pressures Of Chegg
Comments are closed.