Solved Solve Using Dimensional Analysis Chegg
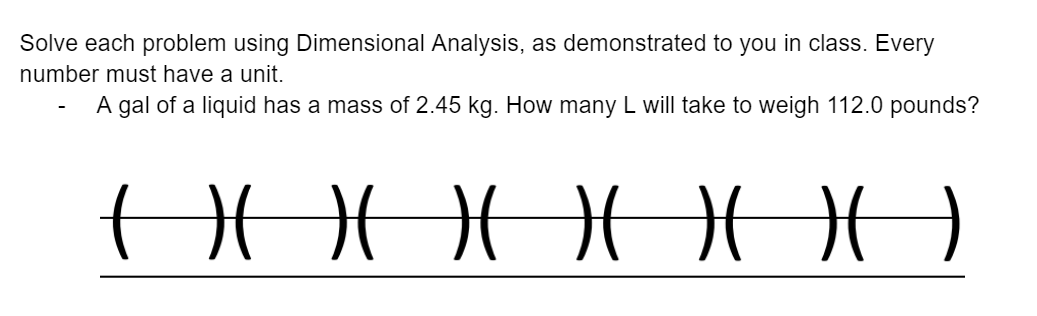
Solved Solve Using Dimensional Analysis Chegg Our expert help has broken down your problem into an easy to learn solution you can count on. there are 2 steps to solve this one. step 1: identify the givens and unknowns. not the question you’re looking for? post any question and get expert help quickly. Write the conversion factors (as ratios) for the number of: (a) kilometers in 1 mile. (b) liters in 1 liquid quart. (c) grams in 1 ounce. the label on a soft drink bottle gives the volume in two units: 2.0 l and 67.6 fl oz. use this information to derive a conversion factor between the english and metric units.

Solve Using Dimensional Analysis Chegg Dt2 l 3. find a proportionality relationship using dimensional analysis of centrifu gal force f in terms of mass m, velocity v and radius r. solution: we set: f = kmavbrc, where k is dimensionless, and compare dimensions:. Master dimensional analysis in chemistry, nursing, and exams with step by step guides, practice problems, and a handy calculator. (i) to convert a physical quantity from one system of units to another. example 1.12. convert 76 cm of mercury pressure into nm−2 using the method of dimensions. solution. in cgs system 76 cm of mercury pressure = 76 × 13.6 × 980 dyne cm−2. the dimensional formula of pressure p is [ml−1t−2] example 1.13. Dimensional analysis explained (part 1) this video explains the fundamental concepts of dimensional analysis and demonstrate how important they are to solving problems.
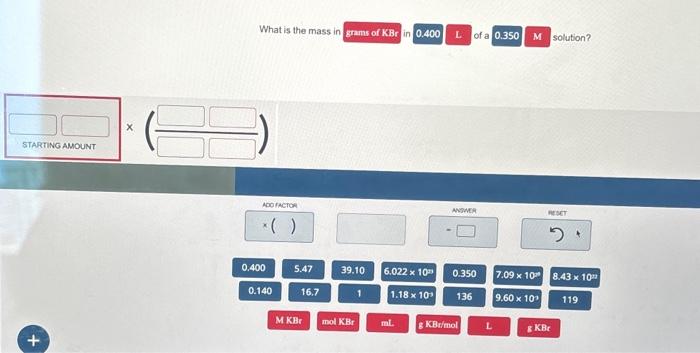
Solved Solve Using Dimensional Analysis Chegg (i) to convert a physical quantity from one system of units to another. example 1.12. convert 76 cm of mercury pressure into nm−2 using the method of dimensions. solution. in cgs system 76 cm of mercury pressure = 76 × 13.6 × 980 dyne cm−2. the dimensional formula of pressure p is [ml−1t−2] example 1.13. Dimensional analysis explained (part 1) this video explains the fundamental concepts of dimensional analysis and demonstrate how important they are to solving problems. To solve a dosage calculation problem using dimensional analysis, you first identify the given quantity and the desired quantity, then set up conversion factors to cancel out unwanted units. When starting to solve a dimensional analysis problem, focus on what the units are for the final answer. example: your car’s average gas mileage is 20 miles gallon and you drive an average of 15,000 miles year. Download exercises dimensional analysis practice problems with answers | university of michigan (um) ann arbor | easy dimensional analysis, harder dimensional analysis, medium dimensional analysis and hard dimensional analysis. This page contains dimensional analysis practice problems for class 11 along with downloadable pdf. practice these problems for better understanding of this topic.

Solved Dimensional Analysis Applications Solve The Following Chegg To solve a dosage calculation problem using dimensional analysis, you first identify the given quantity and the desired quantity, then set up conversion factors to cancel out unwanted units. When starting to solve a dimensional analysis problem, focus on what the units are for the final answer. example: your car’s average gas mileage is 20 miles gallon and you drive an average of 15,000 miles year. Download exercises dimensional analysis practice problems with answers | university of michigan (um) ann arbor | easy dimensional analysis, harder dimensional analysis, medium dimensional analysis and hard dimensional analysis. This page contains dimensional analysis practice problems for class 11 along with downloadable pdf. practice these problems for better understanding of this topic.

Solved Directions Solve Each Problem Using Dimensional Chegg Download exercises dimensional analysis practice problems with answers | university of michigan (um) ann arbor | easy dimensional analysis, harder dimensional analysis, medium dimensional analysis and hard dimensional analysis. This page contains dimensional analysis practice problems for class 11 along with downloadable pdf. practice these problems for better understanding of this topic.
Comments are closed.