Solved Experiment 8 Prelab Chegg
Solved Experiment 8 Prelab Chegg Unlock this question and get full access to detailed step by step answers. question: experiment 8: creating a buffer solution prelab questions 1) what is buffer? give two examples. Two special steps in the experimental procedure are incorporated to reduce the loss of the calcium oxalate precipitate. identify the steps in the procedure and the reason for each step.

Solved Experiment 8 Prelab Assignment Chem 1090 General Chegg The limiting reactant is determined in this experiment. what are the reactants (and their molar masses) in the experiment?. Read through the experimental procedure in chemtrek and watch the associated experiment videos in canvas. in the provided space, write a lab goal, summarize the procedure, record your observations, and complete all questions included in this report sheet. Study with quizlet and memorize flashcards containing terms like limiting reactant cacl2 x 2h2o excess reactant k2c2o4 x h2o, by placing potassium oxalate in and ppt forms calcium chloride would be the excess. Title: experiment 8: limiting reactant objective purpose: to determine the limiting reactant in a mixture of two soluble salts. to determine the percent composition of each substance in a salt mixture. procedure: **part a prepare the salt mixture. a.** mass of salt mixture. measure the mass of beaker 1 and record on the report sheet for trial 1.

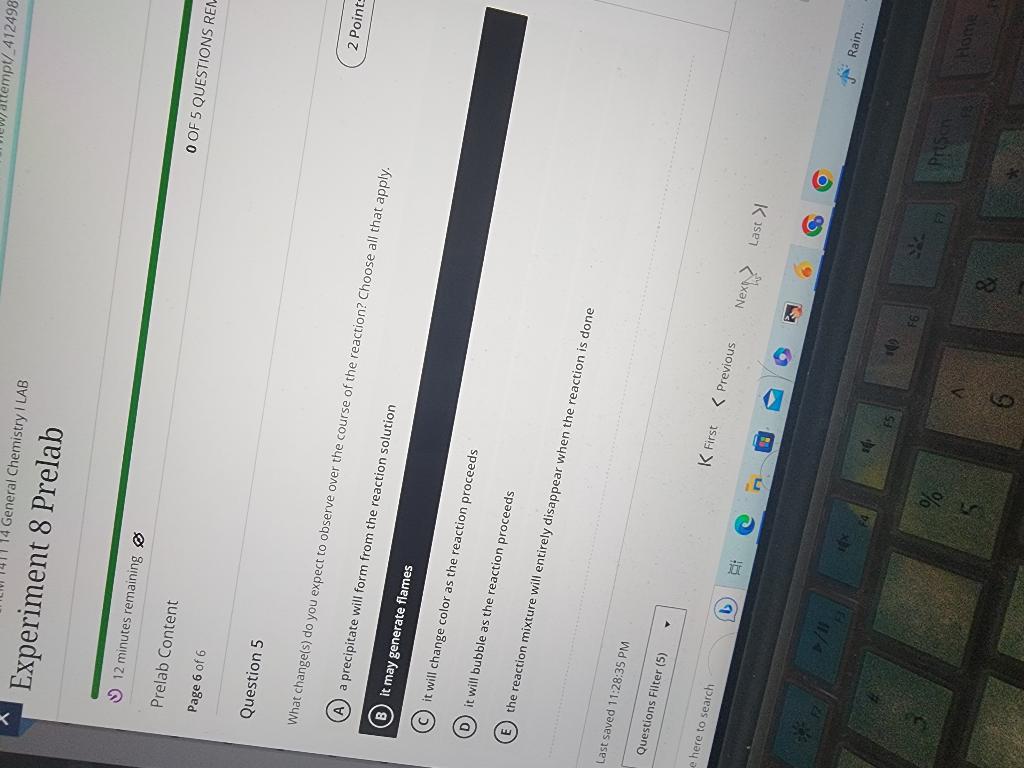
Solved Chemistry 111 Experiment 8 Prelab Activities 1 Road Chegg Study with quizlet and memorize flashcards containing terms like limiting reactant cacl2 x 2h2o excess reactant k2c2o4 x h2o, by placing potassium oxalate in and ppt forms calcium chloride would be the excess. Title: experiment 8: limiting reactant objective purpose: to determine the limiting reactant in a mixture of two soluble salts. to determine the percent composition of each substance in a salt mixture. procedure: **part a prepare the salt mixture. a.** mass of salt mixture. measure the mass of beaker 1 and record on the report sheet for trial 1. Experiment 8 pre lab quiz save describe the four factors that contribute to the rate of a particular chemical reaction click the card to flip 👆. The reactants in this experiment are calcium chloride dihydrate and potassium oxalate monohydrate. the molar mass of calcium chloride dihydrate is 147 g mol. the molar mass of potassium oxalate monohydrate is 184 g mol. the limiting reactant is determined by observing the precipitate, calcium oxalate monohydrate, for an excess of calcium ion. Assume a student performed the acid base titration experiment that you will be doing in expt. 8. the student accurately transferred 5.00 ml of vinegar into an erlenmyer flask, added indicator, and performed the titration by adding sodium hydroxide. Study with quizlet and memorize flashcards containing terms like what are the objectives of the lab?, what are the reactions for this experiment?, what is le chatlier's principle? and more.

Digital Circuits Can You Please Do The Prelab Chegg Experiment 8 pre lab quiz save describe the four factors that contribute to the rate of a particular chemical reaction click the card to flip 👆. The reactants in this experiment are calcium chloride dihydrate and potassium oxalate monohydrate. the molar mass of calcium chloride dihydrate is 147 g mol. the molar mass of potassium oxalate monohydrate is 184 g mol. the limiting reactant is determined by observing the precipitate, calcium oxalate monohydrate, for an excess of calcium ion. Assume a student performed the acid base titration experiment that you will be doing in expt. 8. the student accurately transferred 5.00 ml of vinegar into an erlenmyer flask, added indicator, and performed the titration by adding sodium hydroxide. Study with quizlet and memorize flashcards containing terms like what are the objectives of the lab?, what are the reactions for this experiment?, what is le chatlier's principle? and more.

Solved Experiment 17 Prelab The Glove Experiment Prelab Chegg Assume a student performed the acid base titration experiment that you will be doing in expt. 8. the student accurately transferred 5.00 ml of vinegar into an erlenmyer flask, added indicator, and performed the titration by adding sodium hydroxide. Study with quizlet and memorize flashcards containing terms like what are the objectives of the lab?, what are the reactions for this experiment?, what is le chatlier's principle? and more.
Comments are closed.