Solved Classifying Reactions For Each Reaction Name The Type Chegg

Solved Classifying Reactions For Each Reaction Name The Type Chegg Here’s the best way to solve it. solution : the balanced reactions and their types are given below. Most chemical reactions can be classified into one or more of five basic types: acid–base reactions, exchange reactions, condensation reactions (and the reverse, cleavage reactions), and oxidation–reduction reactions.
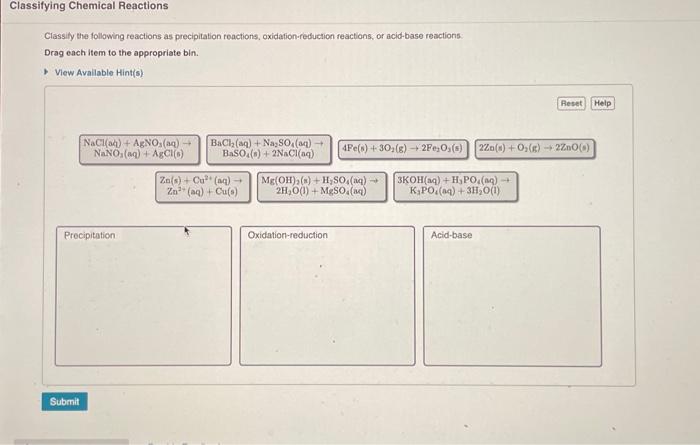
Solved Classifying Chemical Reactions Classily The Following Chegg Text: classify each chemical reaction: type of reaction (check all that apply): combination, precipitation, single replacement, combustion, double replacement, acid base, decomposition. This module will provide an introduction to three of the most prevalent types of chemical reactions: precipitation, acid base, and oxidation reduction. Study with quizlet and memorize flashcards containing terms like classify each of the following chemical reactions. s o2 → so2 cacl2 2agno3 → ca (no3)2 2agcl zn cuso4 → cu znso4 2na2o → 4na o2, ? 2nabr → 2nacl br2 mg ? → mgo h2, 2kclo3 → 2kcl ? 3o2 3cl2 3k and more. Define five common types of chemical reactions (single replacement, double replacement, composition, decomposition, and combustion). classify chemical reactions as one of these five types given appropriate descriptions or chemical equations.

Solved Classifying Chemical Reactions Continued Classifying Chegg Study with quizlet and memorize flashcards containing terms like classify each of the following chemical reactions. s o2 → so2 cacl2 2agno3 → ca (no3)2 2agcl zn cuso4 → cu znso4 2na2o → 4na o2, ? 2nabr → 2nacl br2 mg ? → mgo h2, 2kclo3 → 2kcl ? 3o2 3cl2 3k and more. Define five common types of chemical reactions (single replacement, double replacement, composition, decomposition, and combustion). classify chemical reactions as one of these five types given appropriate descriptions or chemical equations. There are 2 steps to solve this one. to classify the reaction , look for the formation of a precipitate and the exchange of ions between the reactants. two or more reactants combine together to form a single product. a new.
Comments are closed.