Solved Apply Heisenberg S Position Momentum Uncertainty Chegg
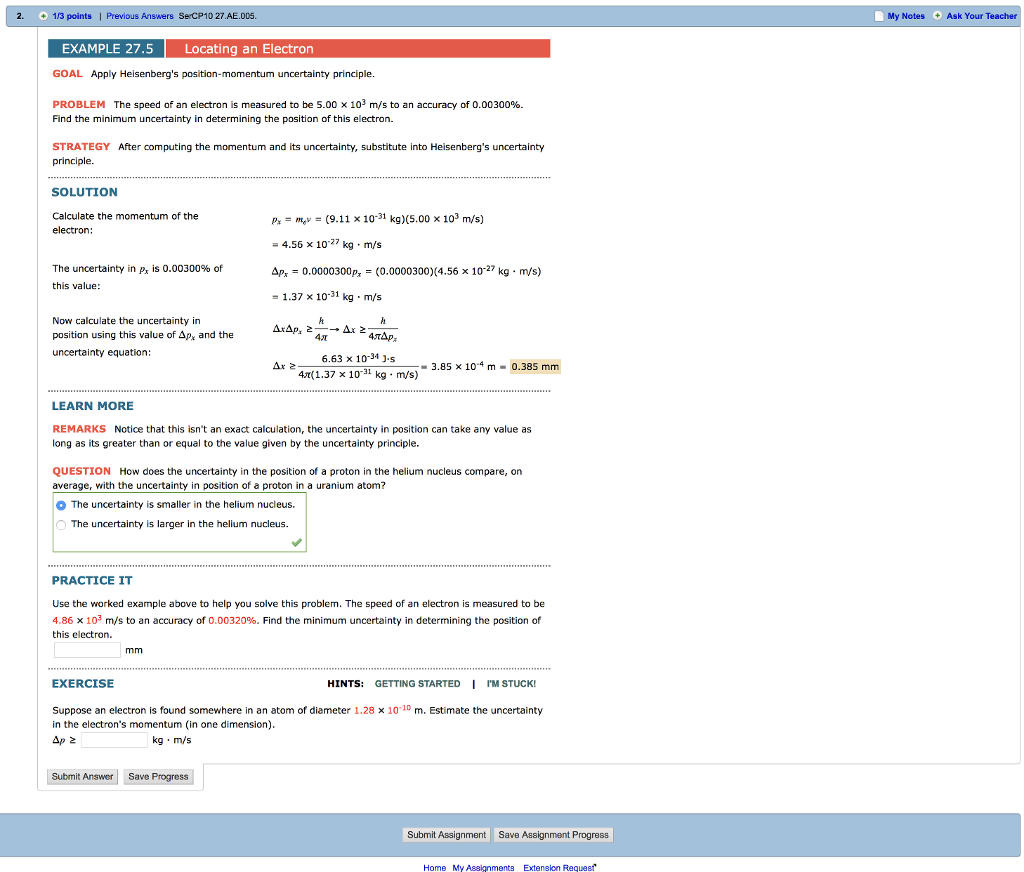
Solved Apply Heisenberg S Position Momentum Uncertainty Chegg Calculate the momentum of the electron using its mass and measured speed. apply heisenberg's position momentum uncertainty principle. the speed of an electron is measured to be 5.00 times 10^3 m s to an accuracy of 0.00300%. find the minimum uncertainty in determining the position of this electron. Heisenberg’s uncertainty principle is a key principle in quantum mechanics. very roughly, it states that if we know everything about where a particle is located (the uncertainty of position is small), we know nothing about its momentum (the uncertainty of momentum is large), and vice versa.

Solved B Use The Heisenberg Momentum Position Uncertainty Chegg It states that there is a limit to the precision with which certain pairs of physical properties, such as position and momentum, can be simultaneously known. in other words, the more accurately one property is measured, the less accurately the other property can be known. According to this principle, it is impossible to determine both the exact position and the exact momentum (velocity × mass) of a moving particle at the same time. if the position of a particle is measured very accurately, then its momentum becomes uncertain. Reproduce known results for the heisenberg's uncertainty principle applied to the position and linear momentum of a particle. understand the difference between compatible and incompatible observables. Heisenberg’s uncertainty principle is a key principle in quantum mechanics. very roughly, it states that if we know everything about where a particle is located (the uncertainty of position is small), we know nothing about its momentum (the uncertainty of momentum is large), and vice versa.

Solved 5 15 Points Heisenberg S Uncertainty Principle Chegg Reproduce known results for the heisenberg's uncertainty principle applied to the position and linear momentum of a particle. understand the difference between compatible and incompatible observables. Heisenberg’s uncertainty principle is a key principle in quantum mechanics. very roughly, it states that if we know everything about where a particle is located (the uncertainty of position is small), we know nothing about its momentum (the uncertainty of momentum is large), and vice versa. We want to define the uncertainty Δa(Ψ) of the hermitian operator a on the state Ψ. this uncertainty should vanish if and only if the state is an eigenstate of a. the uncertainty, moreover, should be a real number. Learn about the heisenberg uncertainty principle in physics. get its definition, simple explanation, formulas, and worked example problems. Werner heisenberg discovered a fundamental rule: you cannot know both the position ($\delta x$) and momentum ($\delta p$) of a particle with infinite precision at the same time. try narrowing the box. notice how the momentum wave packet (Δp) widens!. Heisenberg’s uncertainty principle is a key principle in quantum mechanics. very roughly, it states that if we know everything about where a particle is located (the uncertainty of position is small), we know nothing about its momentum (the uncertainty of momentum is large), and vice versa.

Solved Position And Momentum Are Subject To Heisenbergs Chegg We want to define the uncertainty Δa(Ψ) of the hermitian operator a on the state Ψ. this uncertainty should vanish if and only if the state is an eigenstate of a. the uncertainty, moreover, should be a real number. Learn about the heisenberg uncertainty principle in physics. get its definition, simple explanation, formulas, and worked example problems. Werner heisenberg discovered a fundamental rule: you cannot know both the position ($\delta x$) and momentum ($\delta p$) of a particle with infinite precision at the same time. try narrowing the box. notice how the momentum wave packet (Δp) widens!. Heisenberg’s uncertainty principle is a key principle in quantum mechanics. very roughly, it states that if we know everything about where a particle is located (the uncertainty of position is small), we know nothing about its momentum (the uncertainty of momentum is large), and vice versa.

Solved 1 A Use The Heisenberg Momentum Position Chegg Werner heisenberg discovered a fundamental rule: you cannot know both the position ($\delta x$) and momentum ($\delta p$) of a particle with infinite precision at the same time. try narrowing the box. notice how the momentum wave packet (Δp) widens!. Heisenberg’s uncertainty principle is a key principle in quantum mechanics. very roughly, it states that if we know everything about where a particle is located (the uncertainty of position is small), we know nothing about its momentum (the uncertainty of momentum is large), and vice versa.
Comments are closed.