Solved 1s Chegg

Solved Solve 1a 1t 1s ï For S Chegg There are 3 steps to solve this one. i have created the above part in solidworks as per the given instructions.i have selected the inch u not the question you’re looking for? post any question and get expert help quickly. For example, in the ground state of the hydrogen atom, the single electron is in the 1s orbital, whereas in the first excited state, the atom has absorbed energy and the electron has been promoted to one of the n = 2 orbitals.

Solved Solve 1a 1t 1s ï For S Chegg Solved a graph of the radial probability distribution | chegg image size:700x403 the hydrogen atom the probability distribution of the hydrogen atomimage size:496x396 aleks: interpreting the radial probability distribution of an orbitalimage size:1280x720 figure 1 shows the radial distribution function of a | chegg image size:564x654. Question 02: (a) calculate the probability that an electron described by a hydrogen atomic 1s wavefunction will be found within one bohr radius (a0) of the nucleus. (b) show that the most probable value of r (rmp) in a 1s orbital is 0. Test your knowledge anytime with practice questions. create flashcards from your questions to quiz yourself. ask for examples or analogies of complex concepts to deepen your understanding. polish your papers with expert proofreading and grammar checks. create citations for your assignments in 7,000 styles. An atomic orbital is a function that describes one electron in an atom. the wavefunction with \ (n = 1\), \ (l\) = 0 is called the 1s orbital, and an electron that is described by this function is said to be “in” the ls orbital, i.e. have a 1s orbital state.
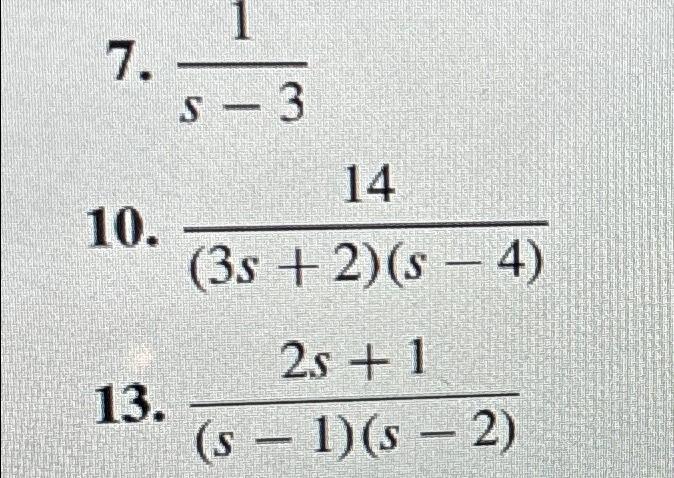
Solved 1s 314 3s 2 S 4 2s 1 S 1 S 2 Chegg Test your knowledge anytime with practice questions. create flashcards from your questions to quiz yourself. ask for examples or analogies of complex concepts to deepen your understanding. polish your papers with expert proofreading and grammar checks. create citations for your assignments in 7,000 styles. An atomic orbital is a function that describes one electron in an atom. the wavefunction with \ (n = 1\), \ (l\) = 0 is called the 1s orbital, and an electron that is described by this function is said to be “in” the ls orbital, i.e. have a 1s orbital state. Calculate the probability of finding hydrogen 1s electron between one and two bohr radius (1a0 –2a0). here’s the best way to solve it. not the question you’re looking for? post any question and get expert help quickly. In this section and in chapter 10 we will explore some of the many other single electron functions that also can be used as atomic orbitals. hydrogenic spin orbitals used as components of multi electron systems are identified in the same way as they are for the hydrogen atom. Enhanced with ai, our expert help has broken down your problem into an easy to learn solution you can count on. here’s the best way to solve it. not the question you’re looking for? post any question and get expert help quickly. Our expert help has broken down your problem into an easy to learn solution you can count on. question: a) calculate for the 1s orbital in the hydrogen atom. b) calculate for the 1s orbital in the hydrogen atom. c) calculate the average potential energy for the 1s orbital. d) compare the result in c) with the total energy.
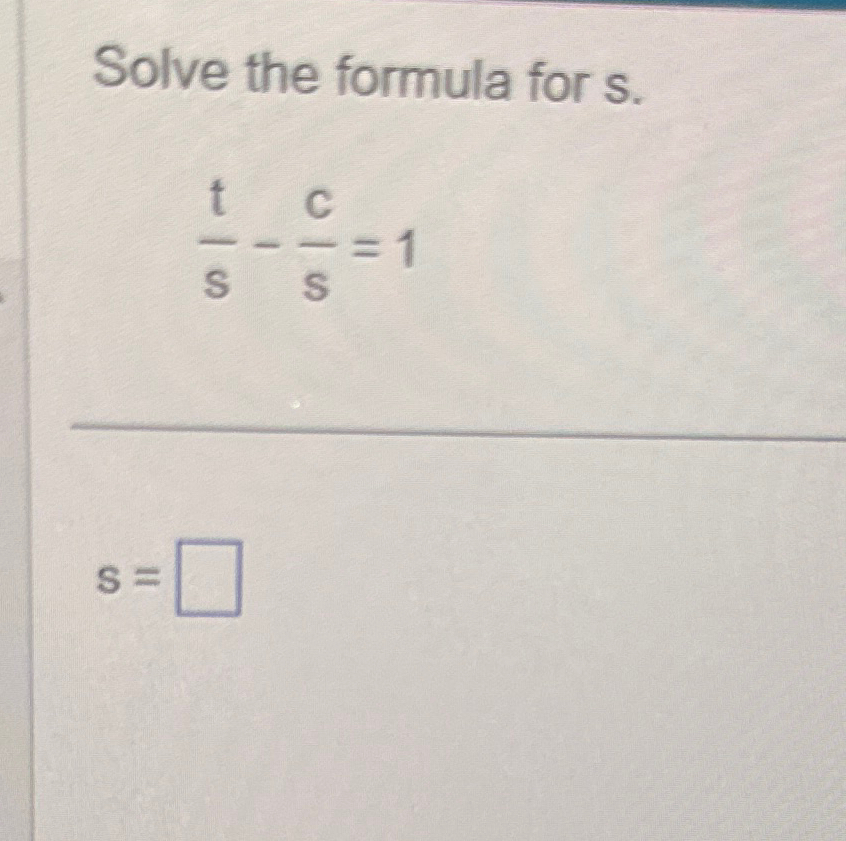
Solved Solve The Formula For S Ts Cs 1s Chegg Calculate the probability of finding hydrogen 1s electron between one and two bohr radius (1a0 –2a0). here’s the best way to solve it. not the question you’re looking for? post any question and get expert help quickly. In this section and in chapter 10 we will explore some of the many other single electron functions that also can be used as atomic orbitals. hydrogenic spin orbitals used as components of multi electron systems are identified in the same way as they are for the hydrogen atom. Enhanced with ai, our expert help has broken down your problem into an easy to learn solution you can count on. here’s the best way to solve it. not the question you’re looking for? post any question and get expert help quickly. Our expert help has broken down your problem into an easy to learn solution you can count on. question: a) calculate for the 1s orbital in the hydrogen atom. b) calculate for the 1s orbital in the hydrogen atom. c) calculate the average potential energy for the 1s orbital. d) compare the result in c) with the total energy.

Solved Solve 1r 1s 1d ï For R Chegg Enhanced with ai, our expert help has broken down your problem into an easy to learn solution you can count on. here’s the best way to solve it. not the question you’re looking for? post any question and get expert help quickly. Our expert help has broken down your problem into an easy to learn solution you can count on. question: a) calculate for the 1s orbital in the hydrogen atom. b) calculate for the 1s orbital in the hydrogen atom. c) calculate the average potential energy for the 1s orbital. d) compare the result in c) with the total energy.
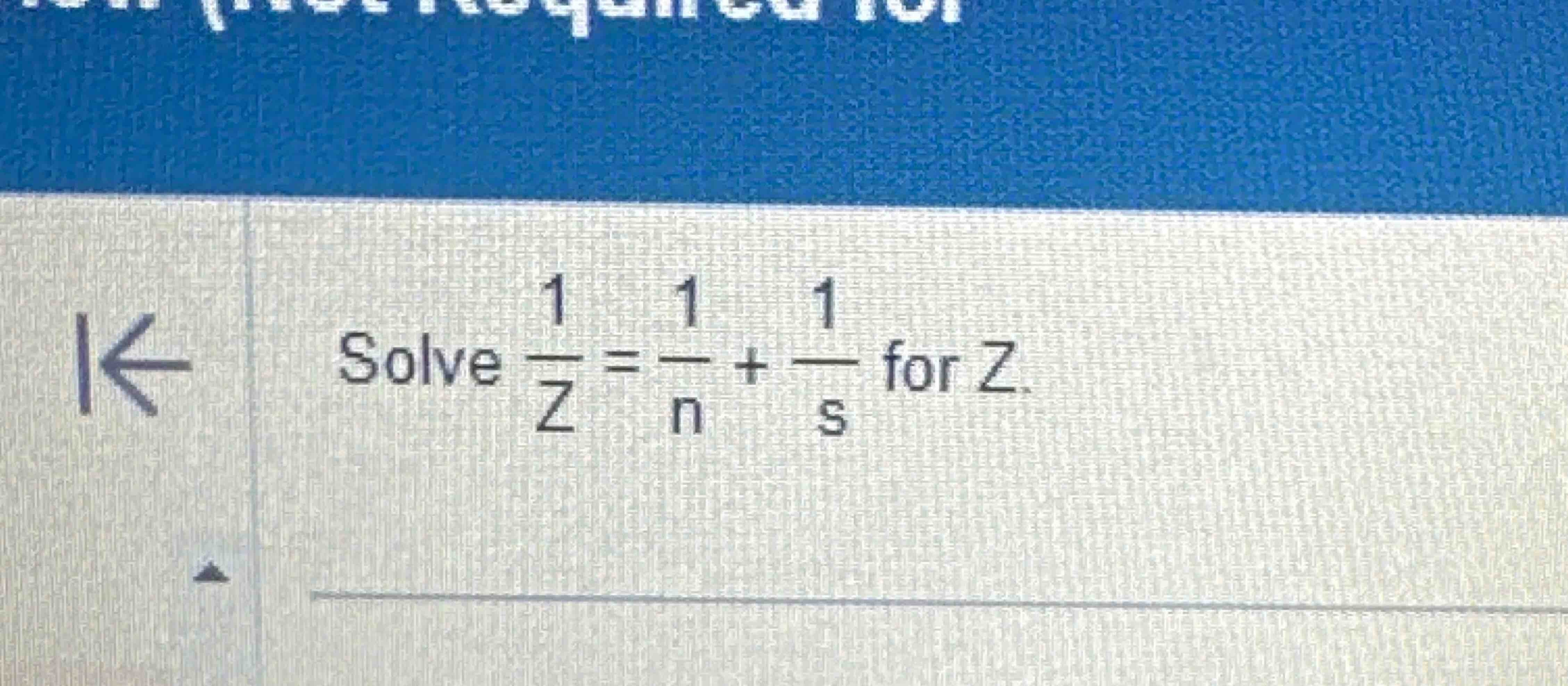
Solved Solve 1z 1n 1s ï For Z Chegg
Comments are closed.