Solution Schrodinger Equation And Wave Function Theory Studypool

Schrödinger Wave Equation Pdf Schrödinger Equation Wave Function In this section, using classical manufacturing, mechanics, functional density concept, and electromagnetic and quantum to model, further contribute to the nanoelectromechanical theory theory we the simulate nanosystems. The time dependent schrödinger equation described above predicts that wave functions can form standing waves, called stationary states. these states are particularly important as their individual study later simplifies the task of solving the time dependent schrödinger equation for any state.
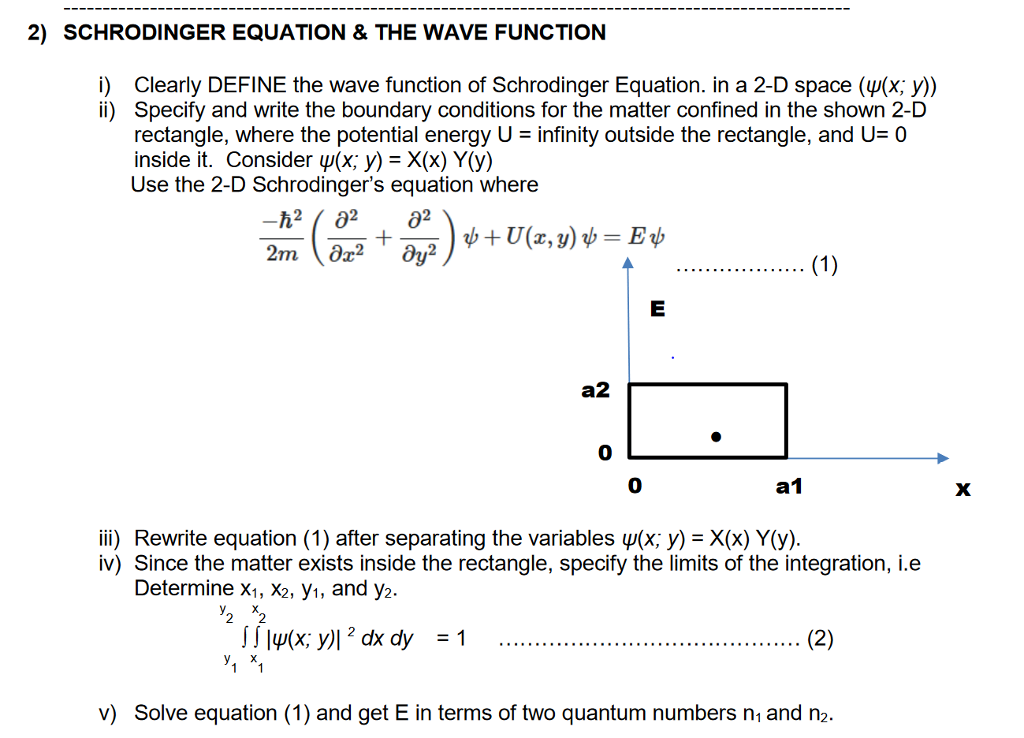
Solved 2 Schrodinger Equation The Wave Function I Chegg Schrödinger’s approach treats electrons as three dimensional standing waves. it is a requirement of standing waves that they be in phase with one another to avoid cancellation; this results in a limited number of solutions (wavefunctions), each of which is associated with a particular energy. We also explore the statistical interpretation of the physical meaning of wave functions obtained as solutions to the schrödinger equation, perspectives on quantum indeterminacy, and the physical meaning of measurement in the copenhagen interpretation (collapse of the wave function). The schrödinger equation is the fundamental equation in quantum mechanics that describes how the wavefunction of a quantum system evolves over time. the wavefunction contains all the information about the system’s physical properties, such as position, momentum, and energy. the schrödinger equation provides a way to predict the future behavior of quantum systems based on their current state. In the first section of this chapter, we discussed the postulates of quantum mechanics i.e. the step by step procedure to solve a quantum mechanical problem. now it’s the time to implement those rules to the simplest quantum mechanical problem i.e. particle in a one dimensional box.

Schrodinger Wave Equation Pdf The schrödinger equation is the fundamental equation in quantum mechanics that describes how the wavefunction of a quantum system evolves over time. the wavefunction contains all the information about the system’s physical properties, such as position, momentum, and energy. the schrödinger equation provides a way to predict the future behavior of quantum systems based on their current state. In the first section of this chapter, we discussed the postulates of quantum mechanics i.e. the step by step procedure to solve a quantum mechanical problem. now it’s the time to implement those rules to the simplest quantum mechanical problem i.e. particle in a one dimensional box. To understand the wave function further, we require a wave equation from which we can study the evolution of wave functions as a function of position and time, in general within a potential field (e.g. the potential fields associated with the coulomb or strong nuclear force). The solution of the schr odinger equation is the wave function (~r;t) which describes the state of a particle moving in the potential u(~r;t). the observable directly linked to the wave function is the probability to nd the particle at position ~rat time t, namely, j (~r;t)j2. Once the potential energy of a particle is specified—or, equivalently, once the force on the particle is specified—we can solve this differential equation for the wave function. Developed and formulated by the austrian physicist erwin schrödinger in 1926, schrodinger’s equation describes the energy and position of an electron in space and time while taking into account the wave matter duality nature of that electron inside the atom.
Comments are closed.