Solution Preparation
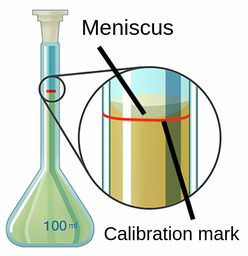
5 Ways To Teach Solution Preparation To Students To prepare a solution that contains a specified concentration of a substance, it is necessary to dissolve the desired number of moles of solute in enough solvent to give the desired final volume of solution. From simple dilutions to complex titrations, the ability to prepare solutions accurately is a fundamental skill. but solution preparation is more than just mixing substances; it’s a delicate dance between precision and understanding, a blend of art and science.

Volumetric Solution Preparation Pdf Preparation of solutions calculator is a useful tool which allows you to calculate how many solid chemicals or stock solutions you will need to prepare the desired solution. Learn how to prepare solutions for high school science laboratory with basic concepts, recipes, and tips. find out how to calculate molarity, percent solutions, and conversion between them. It provides definitions, calculations, and examples for accurate solution preparation, emphasizing the importance of using correct weights and adjusting for hydrated or anhydrous forms. Learn accurate solution preparation, concentration calculation, volume measurement, and dilution techniques for lab experiments.

Lab Datapreparation Of Stock Solutionpreparation Of Chegg It provides definitions, calculations, and examples for accurate solution preparation, emphasizing the importance of using correct weights and adjusting for hydrated or anhydrous forms. Learn accurate solution preparation, concentration calculation, volume measurement, and dilution techniques for lab experiments. Prepare by mixing the volumes of colorimetric solutions and hydrochloric acid (1 percent w v hcl) as indicated in table 2. note – reference solutions must be prepared immediately before use from the colorimetric solutions which may be stored in the refrigerator. Standard solution preparation a standard solution is a solution of known concentration used for calibration in quantitative analysis, such as titrations or spectrophotometric assays. There are several ways of expressing solution concentrations such as moles per litre, percentage concentration, grams per litre, saturated solutions etc. in a school science laboratory there are 5 main methods used for the calculation of and preparation of solutions. Learn what a standard solution is, why it’s important and how to prepare standard solutions using the dilution and weighing methods.

Preparation Of A Solution Stock Photo Image Of Test 137948378 Prepare by mixing the volumes of colorimetric solutions and hydrochloric acid (1 percent w v hcl) as indicated in table 2. note – reference solutions must be prepared immediately before use from the colorimetric solutions which may be stored in the refrigerator. Standard solution preparation a standard solution is a solution of known concentration used for calibration in quantitative analysis, such as titrations or spectrophotometric assays. There are several ways of expressing solution concentrations such as moles per litre, percentage concentration, grams per litre, saturated solutions etc. in a school science laboratory there are 5 main methods used for the calculation of and preparation of solutions. Learn what a standard solution is, why it’s important and how to prepare standard solutions using the dilution and weighing methods.

Solution Preparation Archives Biology Notes Online There are several ways of expressing solution concentrations such as moles per litre, percentage concentration, grams per litre, saturated solutions etc. in a school science laboratory there are 5 main methods used for the calculation of and preparation of solutions. Learn what a standard solution is, why it’s important and how to prepare standard solutions using the dilution and weighing methods.

Solution Preparation Archives Biology Notes Online
Comments are closed.