Solution Measurements Quantities And Errors Studypool

Solution Measurements Quantities And Errors Studypool A given quantity measurement is essentially result of a comparison between the quantity and thepredefined standard. if two quantities are compared , the result is expressed as a numerical value. We try to reduce error by carrying out additional measurements or by changing our experimental apparatus, but we can never eliminate error altogether. thus, error is inherent in measurement.

Solution Physics Measurements Introduction To Physics Physical It is, therefore, necessary to study errors in measurements and understand how these errors affect values that are computed using measured quantities. by knowing these errors and their effects, one can say something about the reliability and validity of the results of the experiment. Measuring a physical quantity involves comparing the quantity with a reference standard called the unit of the quantity. some physical quantities are taken as base quantities and other quantities are expressed in terms of the base quantities called derived quantities. The instrument serves as an extension of human faculties and in many cases enables a person to determine the value of an unknown quantity which his unaided human faculties could not measure. 3 20 2022 measurements and errors purpose: the main purpose of this experiment is to work on metal cylinder and calculate its volume and density by measuring its mass and dimensions. i will estimate the error those quantities from the measurement and error in the mass and dimensions.
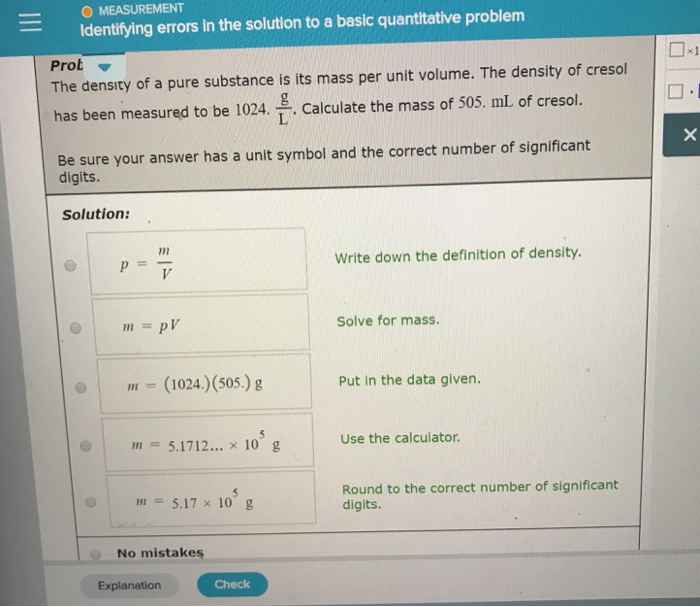
Solved O Measurement Identifying Errors In The Solution To A Chegg Precision the closeness of agreement between independent measurements obtained under the same conditions. it depends only on the distribution of random errors (i.e. the spread of measurements) and does not relate to the true value. Each student uses solid sodium hydroxide, naoh, to prepare a solution of concentration 0.140 mol dm−3. calculate the mass, in grams, of solid sodium hydroxide that each student should weigh out to prepare 250.0 cm3 of a 0.140 mol dm−3 solution. Accuracy is the closeness of agreement between a measured value and a true or accepted value. measurement error is the amount of inaccuracy. precision is a measure of how well a result can be determined (without reference to a theoretical or true value). The document contains daily practice problems (dpp) for neet focusing on measurements and errors. it includes various calculations related to density, percentage errors, significant figures, and formulas for different physical quantities.
Comments are closed.