Solution Halogenation Of Alkenes Studypool

Halogenation Of Alkenes Flashcards Quizlet This page looks at the reaction of the carbon carbon doublebond in alkenes such as ethene with halogens such as chlorine,. A common test is the decolourization of a reddish brown bromine solution by an alkene. the two step mechanism shown in the libretext pages gives you an idea of how the reaction between an alkene and a halogen occurs.
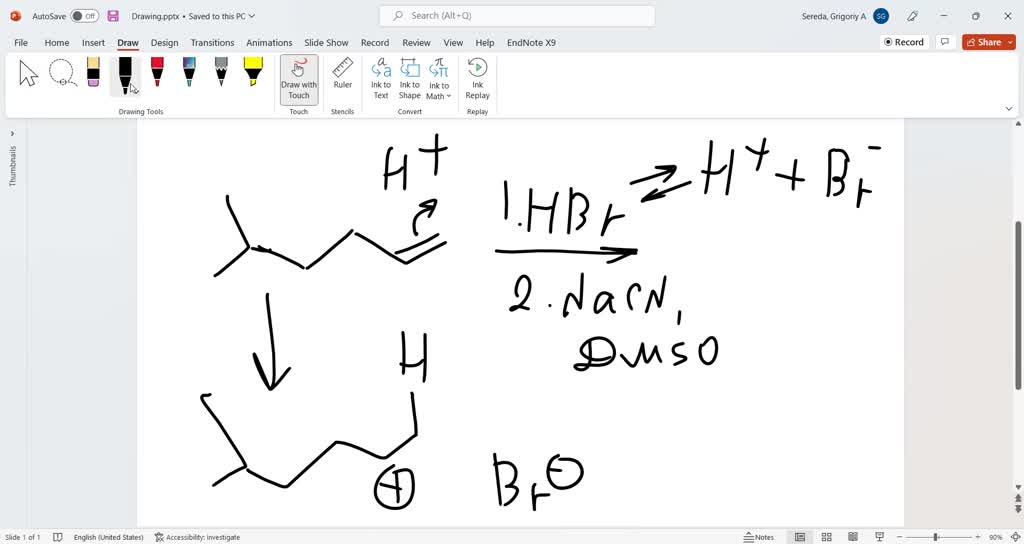
Solved Halogenation Of Alkenes Experiment Discussion By joining chemistry steps, you will gain instant access to the answers and solutions for all the practice problems, including over 40 hours of problem solving videos, multiple choice quizzes, puzzles, reaction maps, and the powerful set of organic chemistry 1 and 2 summary study guides. Measure out 0:5 cm3 of bromine into a hydrocarbon solvent in a test tube. add a few drops of the alkene to the test tube and observe the colour change. Bromine and chlorine add rapidly to alkenes to yield 1,2 dihalides, a process called halogenation. for example, nearly 50 million tons of 1,2 dichloroethane (ethylene dichloride) are synthesized worldwide each year, much of it by addition of cl2 to ethylene. Reaction overview: the alkene halogenation reaction, specifically bromination or chlorination, is one in which a dihalide such as cl2 or br2 is added to a molecule after breaking the carbon to carbon double bond.

Halogenation Of Alkenes Artofit Bromine and chlorine add rapidly to alkenes to yield 1,2 dihalides, a process called halogenation. for example, nearly 50 million tons of 1,2 dichloroethane (ethylene dichloride) are synthesized worldwide each year, much of it by addition of cl2 to ethylene. Reaction overview: the alkene halogenation reaction, specifically bromination or chlorination, is one in which a dihalide such as cl2 or br2 is added to a molecule after breaking the carbon to carbon double bond. A common test is the decolourization of a reddish brown bromine solution by an alkene. the two step mechanism shown in the libretext pages gives you an idea of how the reaction between an alkene and a halogen occurs. In this tutorial, i want to talk about the halogenation of alkenes, which is a signature reaction of alkenes that has all the components of a potential perfect disaster. Bromine and chlorine add rapidly to alkenes to yield 1,2 dihalides, a process called halogenation. for example, nearly 50 million tons of 1,2 dichloroethane (ethylene dichloride) are synthesized worldwide each year, much of it by addition of cl 2 to ethylene. Master the halogenation of an alkene. discover the cyclic halonium ion mechanism, anti addition stereochemistry, and industrial uses in this expert guide.

The Halogenation Addition Of Alkenes Download Scientific Diagram A common test is the decolourization of a reddish brown bromine solution by an alkene. the two step mechanism shown in the libretext pages gives you an idea of how the reaction between an alkene and a halogen occurs. In this tutorial, i want to talk about the halogenation of alkenes, which is a signature reaction of alkenes that has all the components of a potential perfect disaster. Bromine and chlorine add rapidly to alkenes to yield 1,2 dihalides, a process called halogenation. for example, nearly 50 million tons of 1,2 dichloroethane (ethylene dichloride) are synthesized worldwide each year, much of it by addition of cl 2 to ethylene. Master the halogenation of an alkene. discover the cyclic halonium ion mechanism, anti addition stereochemistry, and industrial uses in this expert guide.
Comments are closed.