Solution Bohrs Model For Hydrogen Atom Studypool

The Bohr Model Of The Hydrogen Atom Explained Pdf Energy Level Bohr model of hydrogen atom lesson 5 bohr atom • bohr suggested a model of hydrogen atom that predicted existence of line spectra • bohr used planck's and einstein's idea about quantized e and proposed theme postulates • hydrogen atom has certain allowable e levels which bohr called stationary states. Video answers for all textbook questions of chapter 8, bohr's model of the hydrogen atom, the physics of atoms and quanta by numerade.
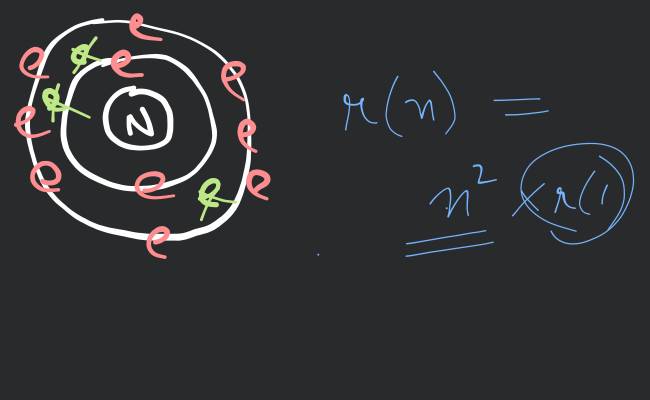
Bohrs Model Of Hydrogen Atom Filo Ncert class 11 chemistry 2.4: bohr’s model for hydrogen atom – quantized orbits, angular momentum, energy levels, and how bohr explained hydrogen’s line spectrum with limitations. Bohr’s model of the hydrogen atom, proposed by niels bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. bohr’s model combines the classical mechanics of planetary motion with the quantum concept of photons. An electron in a bohr hydrogen atom has energy of 2.42* 10^ ( 19)j. the value of n for this electron is: (arada 2nd round model 2023) a 5 b 2 c 3 d 4. Bohr’s model of the hydrogen atom, proposed by niels bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. bohr’s model combines the classical mechanics of planetary motion with the quantum concept of photons.

Solution Bohrs Model For Hydrogen Atom Studypool An electron in a bohr hydrogen atom has energy of 2.42* 10^ ( 19)j. the value of n for this electron is: (arada 2nd round model 2023) a 5 b 2 c 3 d 4. Bohr’s model of the hydrogen atom, proposed by niels bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. bohr’s model combines the classical mechanics of planetary motion with the quantum concept of photons. Mcq practice test & solutions: test: electromagnetic radiation, bohr model (20 questions) you can prepare effectively for jee chemistry for jee main & advanced with this dedicated mcq practice test (available with solutions) on the important topic of " test: electromagnetic radiation, bohr model ". Problem 4.4: find the frequency of revolution of the electron in the classical model of the hydrogen atom. in what region of the spectrum are electromagnetic waves of this frequency? solution: follow the section 4.2. Solutions answers to exercises exercise 1. calculate the energy of the electron in the hydrogen atom if the electron is excited from n = 2 to n = 6. Solve important jee advanced pyqs from bohr model with clear concepts and detailed explanation. a helpful video for revision, concept clarity, and advanced level practice.

Solution Bohrs Model For Hydrogen Atom Studypool Mcq practice test & solutions: test: electromagnetic radiation, bohr model (20 questions) you can prepare effectively for jee chemistry for jee main & advanced with this dedicated mcq practice test (available with solutions) on the important topic of " test: electromagnetic radiation, bohr model ". Problem 4.4: find the frequency of revolution of the electron in the classical model of the hydrogen atom. in what region of the spectrum are electromagnetic waves of this frequency? solution: follow the section 4.2. Solutions answers to exercises exercise 1. calculate the energy of the electron in the hydrogen atom if the electron is excited from n = 2 to n = 6. Solve important jee advanced pyqs from bohr model with clear concepts and detailed explanation. a helpful video for revision, concept clarity, and advanced level practice.
Comments are closed.