Solution Bohrs Atom Model Chemistry Studypool
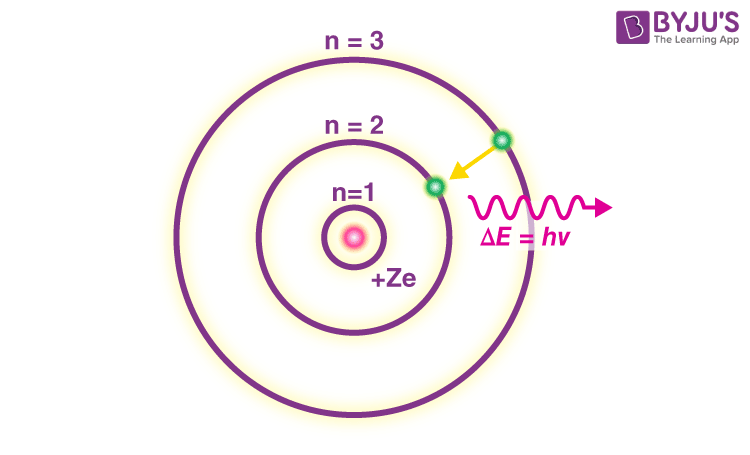
Bohr S Model Of An Atom With Postulates And Limitations Of Bohr S Model Stuck on a study question? our verified tutors can answer all questions, from basic math to advanced rocket science! samson, 1674 gives the illusion of a quintessential quatrain poem with a regular rhyme scheme and the expected generous us. In 1913, a danish physicist, niels bohr (1885–1962; nobel prize in physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum.

Solution Bohrs Model Of Atom Studypool In 1913, niels bohr attempted to resolve the atomic paradox by ignoring classical electromagnetism’s prediction that the orbiting electron in hydrogen would continuously emit light. How do electrons move according to bohr’s model? the theory notes that electrons in atoms travel around a central nucleus in circular orbits and can only orbit stably at a distinct set of distances from the nucleus in certain fixed circular orbits. However, because of its simplicity, and its correct results for selected systems (see below for application), the bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. In this theory, bohr postulated that the electrons in an atom orbit the nucleus in well defined circular paths, known as stationary orbits. this paper aims to explain the key features of bohr's theory and its implications for our understanding of atomic structure and behavior.

Solution Bohrs Model Of Atom And Electro Studypool However, because of its simplicity, and its correct results for selected systems (see below for application), the bohr model is still commonly taught to introduce students to quantum mechanics or energy level diagrams before moving on to the more accurate, but more complex, valence shell atom. In this theory, bohr postulated that the electrons in an atom orbit the nucleus in well defined circular paths, known as stationary orbits. this paper aims to explain the key features of bohr's theory and its implications for our understanding of atomic structure and behavior. Bohr atomic model (1913) the bohr model, proposed by niels bohr, was a major improvement over rutherford’s model. it introduced quantized energy levels for electrons, explaining the stability of atoms and the discrete spectral lines observed in hydrogen. User generated content is uploaded by users for the purposes of learning and should be used following studypool's honor code & terms of service. stuck on a study question? our verified tutors can answer all questions, from basic math to advanced rocket science! brief exercise 5 3 prepare the journal entries to record the fol. Access 20 million homework answers, class notes, and study guides in our notebank. Write the main postulates of bohr's theory and what are its limitations? how this model helped in calculating the energy of an electron in different energy levels of hydrogen atom? ans: bohr retained the essential features of the rutherford model of the atom.
Comments are closed.