Solute Solvent Solution Solubility Chemistry
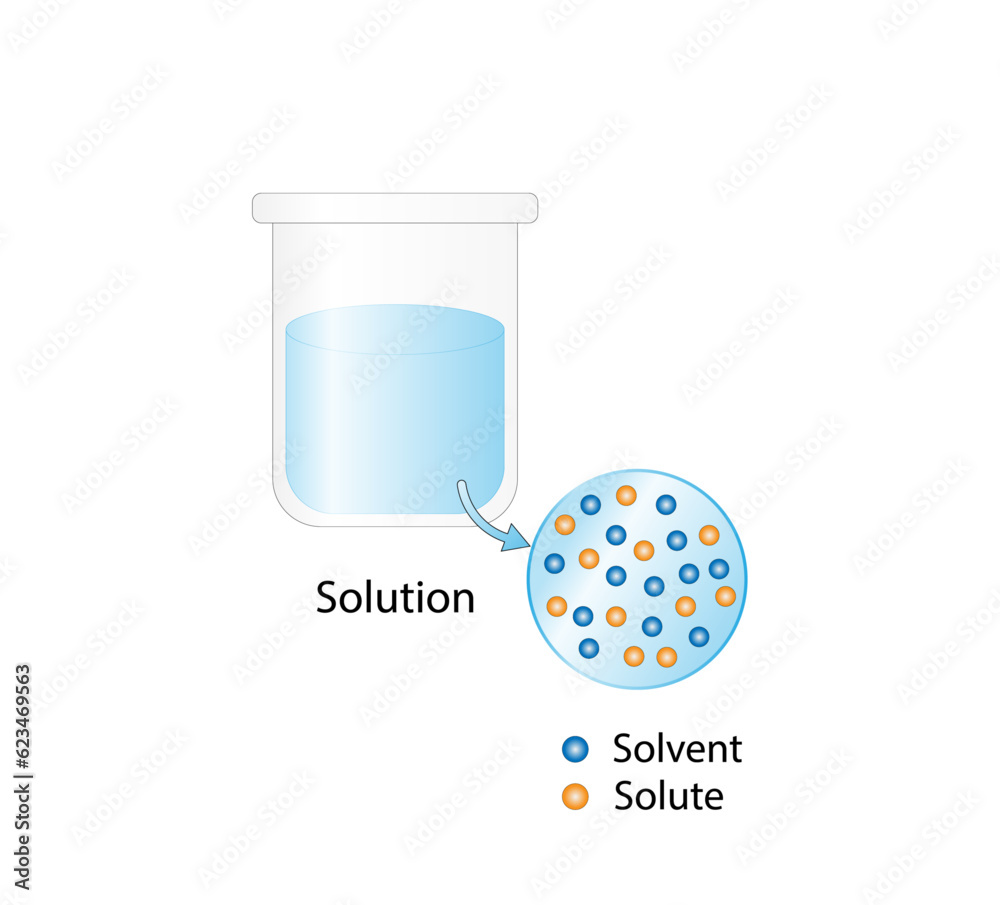
Solution Solubility Homogeneous Mixture Solute Solvent And Solution The solubility of a solute in a particular solvent is the maximum concentration that may be achieved under given conditions when the dissolution process is at equilibrium. when a solute’s concentration is equal to its solubility, the solution is said to be saturated with that solute. The solute does not have to be in the same physical state as the solvent, but the physical state of the solvent usually determines the state of the solution. as long as the solute and solvent combine to give a homogeneous solution, the solute is said to be soluble in the solvent.
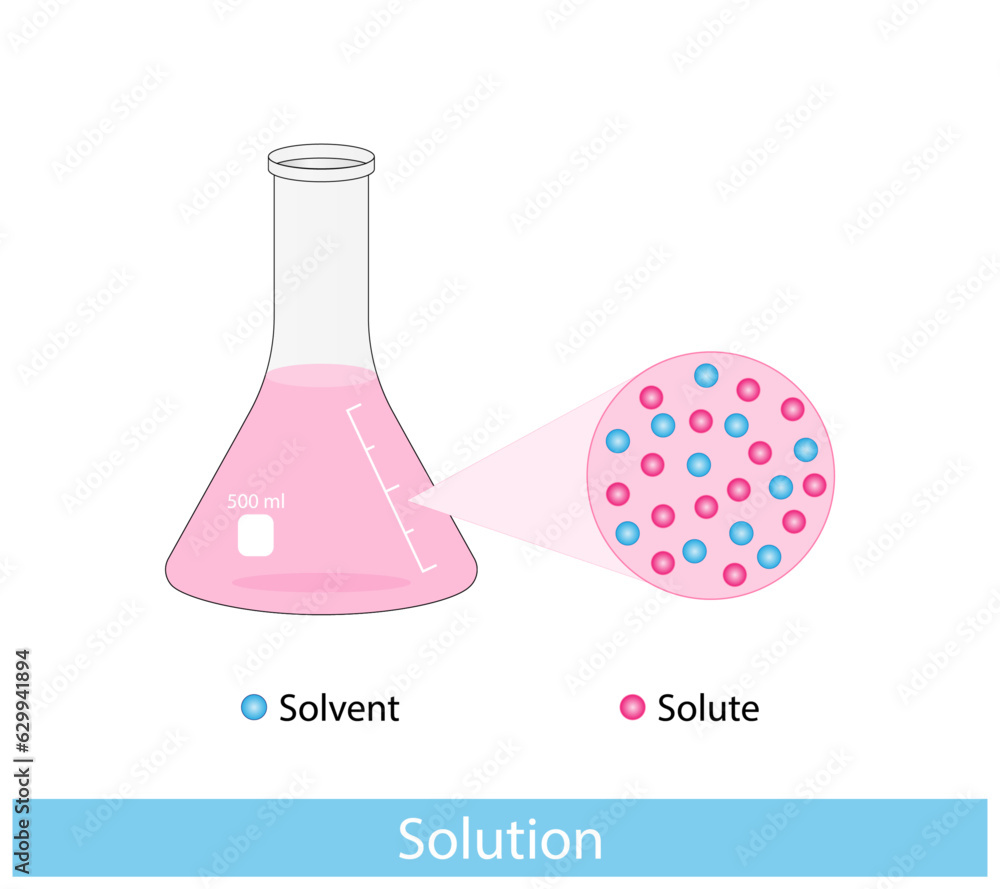
Solution Solubility Homogeneous Mixture Solute Solvent And Solution As for any solution, the solubility of a gas in a liquid is affected by the intermolecular attractive forces between solute and solvent species. When a substance (solute) dissolves, its particles separate and disperse evenly throughout the dissolving medium (solvent). only substances that are soluble in a particular solvent can dissolve and form solutions. Solubility is defined as the upper limit of solute that can be dissolved in a given amount of solvent at equilibrium. in such an equilibrium, le chatelier's principle can be used to explain most of the main factors that affect solubility. Solubility, degree to which a substance dissolves in a solvent to make a solution (usually expressed as grams of solute per litre of solvent). solubility of one fluid (liquid or gas) in another may be complete (totally miscible; e.g., methanol and water) or partial (oil and water dissolve only.
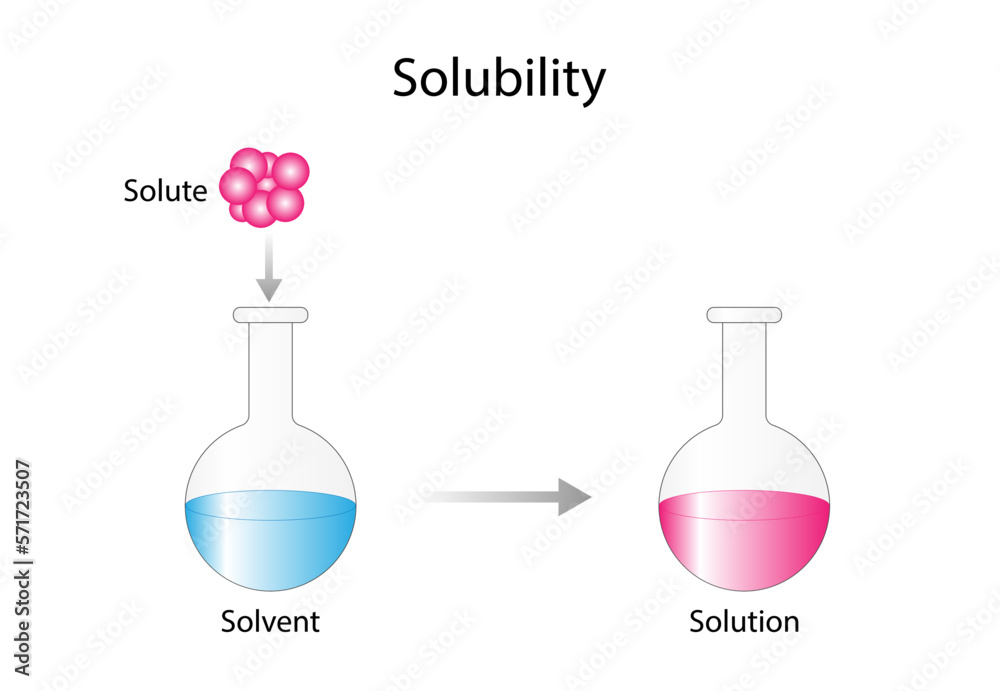
Solutions Solubility Homogeneous Mixture Solute Solvent And Solution Solubility is defined as the upper limit of solute that can be dissolved in a given amount of solvent at equilibrium. in such an equilibrium, le chatelier's principle can be used to explain most of the main factors that affect solubility. Solubility, degree to which a substance dissolves in a solvent to make a solution (usually expressed as grams of solute per litre of solvent). solubility of one fluid (liquid or gas) in another may be complete (totally miscible; e.g., methanol and water) or partial (oil and water dissolve only. In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. insolubility is the opposite property, the inability of the solute to form such a solution. Solubility is the measure of how well a solute can dissolve in a solvent to form a solution. it dictates how substances dissolve and interact, impacting fields like pharmaceuticals, environmental science, cooking, and chemical reactions. Iupac defines solubility as the analytical composition of a saturated solution expressed as a proportion of a designated solute in a designated solvent. solubility may be stated in units of concentration, molality, mole fraction, mole ratio, and other units. The solubility of a solute in a particular solvent is the maximum concentration that may be achieved under given conditions when the dissolution process is at equilibrium. when a solute’s concentration is equal to its solubility, the solution is said to be saturated with that solute.

Solutions Solubility Homogeneous Mixture Solute Solvent And Solution In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. insolubility is the opposite property, the inability of the solute to form such a solution. Solubility is the measure of how well a solute can dissolve in a solvent to form a solution. it dictates how substances dissolve and interact, impacting fields like pharmaceuticals, environmental science, cooking, and chemical reactions. Iupac defines solubility as the analytical composition of a saturated solution expressed as a proportion of a designated solute in a designated solvent. solubility may be stated in units of concentration, molality, mole fraction, mole ratio, and other units. The solubility of a solute in a particular solvent is the maximum concentration that may be achieved under given conditions when the dissolution process is at equilibrium. when a solute’s concentration is equal to its solubility, the solution is said to be saturated with that solute.
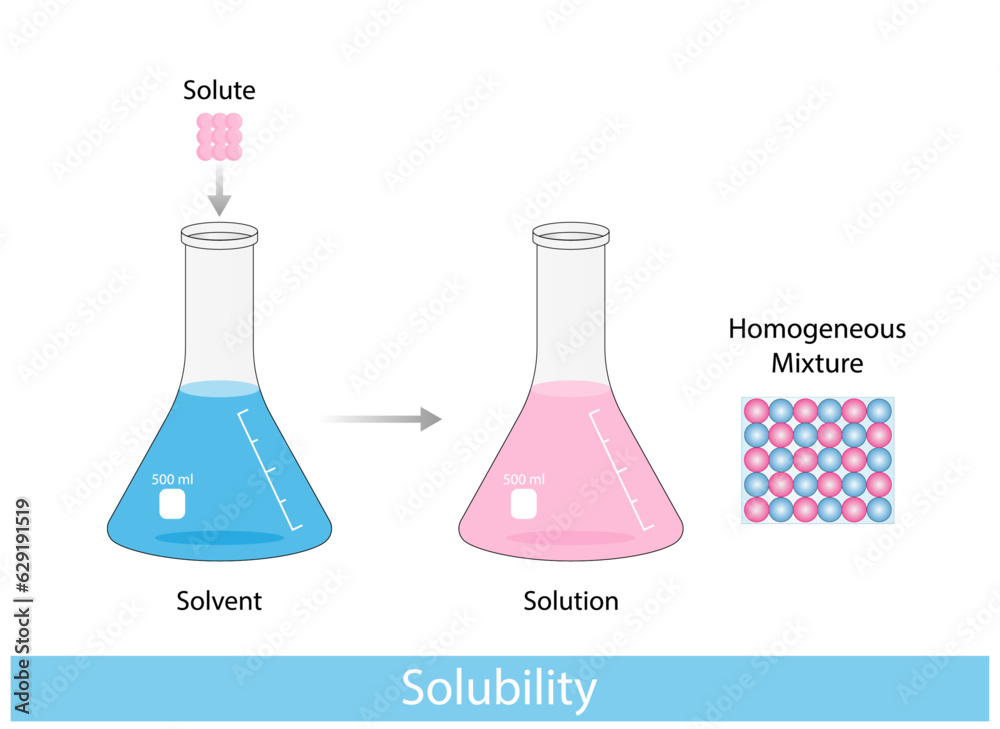
Solutions Solubility Homogeneous Mixture Solute Solvent And Solution Iupac defines solubility as the analytical composition of a saturated solution expressed as a proportion of a designated solute in a designated solvent. solubility may be stated in units of concentration, molality, mole fraction, mole ratio, and other units. The solubility of a solute in a particular solvent is the maximum concentration that may be achieved under given conditions when the dissolution process is at equilibrium. when a solute’s concentration is equal to its solubility, the solution is said to be saturated with that solute.
Comments are closed.