Solubility And Temperature

Ppt Solubility Vs Temperature Powerpoint Presentation Free Download The solubility of the majority of solid substances increases as the temperature increases. however, the effect is difficult to predict and varies widely from one solute to another. As the temperature of a liquid increases, the solubilities of gases in that liquid decrease. we can use the second law of thermodynamics to explain why. heating a solution of a gas enables the particles of gas to move more freely between the solution and the gas phase.

Solubility Curves Solubility Vs Temperature For Solids 140 Learn how temperature and pressure affect the solubility of solids and gases: why heating often increases solubility of solids but decreases gas solubility, and how pressure plays a role. The solubility of the majority of solid substances increases as the temperature increases. however, the effect is difficult to predict and varies widely from one solute to another. The solubility of the majority of solid substances increases as the temperature increases. however, the effect is difficult to predict and varies widely from one solute to another. Explore the relationship between temperature and solubility, examining how different factors like the nature of solute and solvent, and heat of solution, influence this dependency.
Solved The Graph Below Shows How Solubility Changes With Temperature The solubility of the majority of solid substances increases as the temperature increases. however, the effect is difficult to predict and varies widely from one solute to another. Explore the relationship between temperature and solubility, examining how different factors like the nature of solute and solvent, and heat of solution, influence this dependency. The solubility mainly depends on the composition of solute and solvent (including their ph and the presence of other dissolved substances) as well as on temperature and pressure. Learn why temperature affects solubility differently for solids and gases, governed by dissolution energy and underlying thermodynamic principles. However, the solubility of a solute changes as the temperature of the water changes. the solubility of a substance in water at different temperatures can be displayed using a solubility. Learn how temperature and pressure affect the solubility of different types of solutes. find out the definition, examples and applications of solubility and henry's law.
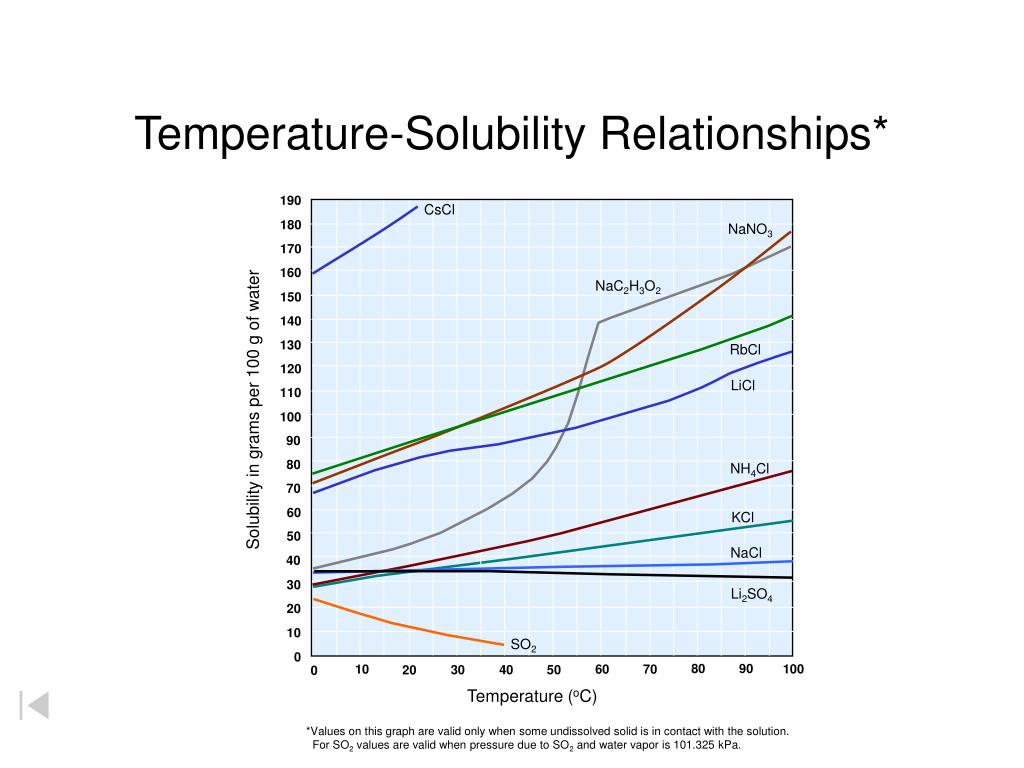
Ppt Solubility Vs Temperature Powerpoint Presentation Free Download The solubility mainly depends on the composition of solute and solvent (including their ph and the presence of other dissolved substances) as well as on temperature and pressure. Learn why temperature affects solubility differently for solids and gases, governed by dissolution energy and underlying thermodynamic principles. However, the solubility of a solute changes as the temperature of the water changes. the solubility of a substance in water at different temperatures can be displayed using a solubility. Learn how temperature and pressure affect the solubility of different types of solutes. find out the definition, examples and applications of solubility and henry's law.
Comments are closed.