Solid Oxide Fuel Cells Pptx

Advancements And Challenges In Solid Oxide Fuel Cells Pptx The document presents an overview of solid oxide fuel cells (sofcs) as a clean and efficient energy source necessary due to the depletion of non renewable energy resources. Sofc free download as powerpoint presentation (.ppt .pptx), pdf file (.pdf), text file (.txt) or view presentation slides online. solid oxide fuel cells (sofcs) operate at high temperatures around 1000°c and use a solid oxide material as the electrolyte to conduct oxygen ions.

Ppt Solid Oxide Fuel Cells Powerpoint Presentation Free Download Current density distribution in a solid oxide fuel cell. comsol. introduction. this example studies the current density distribution in a solid oxide fuel cell (sofc). Discover the innovative technology of solid oxide fuel cells, which offer high efficiency and low emissions. learn about their applications, benefits, and advancements in energy production, including their role in sustainable power generation and hydrogen fuel integration. Solid oxide fuel cells (sofcs) are the second most developed fuel cell technology, ideal for power generation in utilities. these cells utilize ceramic plates packed in multilayer disks. It consists of an anode and cathode separated by an electrolyte, and produces electricity through an electrochemical reaction without combustion. sofcs operate at high temperatures between 1000 1800 degrees f, which allows them to use a wide variety of fuels.

Solid Oxide Fuel Cells Docslib Solid oxide fuel cells (sofcs) are the second most developed fuel cell technology, ideal for power generation in utilities. these cells utilize ceramic plates packed in multilayer disks. It consists of an anode and cathode separated by an electrolyte, and produces electricity through an electrochemical reaction without combustion. sofcs operate at high temperatures between 1000 1800 degrees f, which allows them to use a wide variety of fuels. Doe believes fuel cells have a role in achieving the nation’s clean energy goals but the current state of sofc requires additional basic r&d to mature the technology. This document provides information about different types of fuel cells. it begins by defining a fuel cell as an electrochemical device that converts chemical energy directly into electrical energy. A solid oxide fuel cell (sofc) can be fed with several types of fuel, including hydrogen gas, carbon monoxide and methane, which are direct oxidation fuels within the cell. A solid oxide fuel cell (sofc) uses a hard ceramic electrolyte instead of a liquid and operates at temperatures up to 1,000 degrees c (about 1,800 degrees f). a mixture of zirconium oxide and calcium oxide form a crystal lattice, though other oxide combinations have also been used as electrolytes.

Solid Oxide Fuel Cells Pptx Doe believes fuel cells have a role in achieving the nation’s clean energy goals but the current state of sofc requires additional basic r&d to mature the technology. This document provides information about different types of fuel cells. it begins by defining a fuel cell as an electrochemical device that converts chemical energy directly into electrical energy. A solid oxide fuel cell (sofc) can be fed with several types of fuel, including hydrogen gas, carbon monoxide and methane, which are direct oxidation fuels within the cell. A solid oxide fuel cell (sofc) uses a hard ceramic electrolyte instead of a liquid and operates at temperatures up to 1,000 degrees c (about 1,800 degrees f). a mixture of zirconium oxide and calcium oxide form a crystal lattice, though other oxide combinations have also been used as electrolytes.
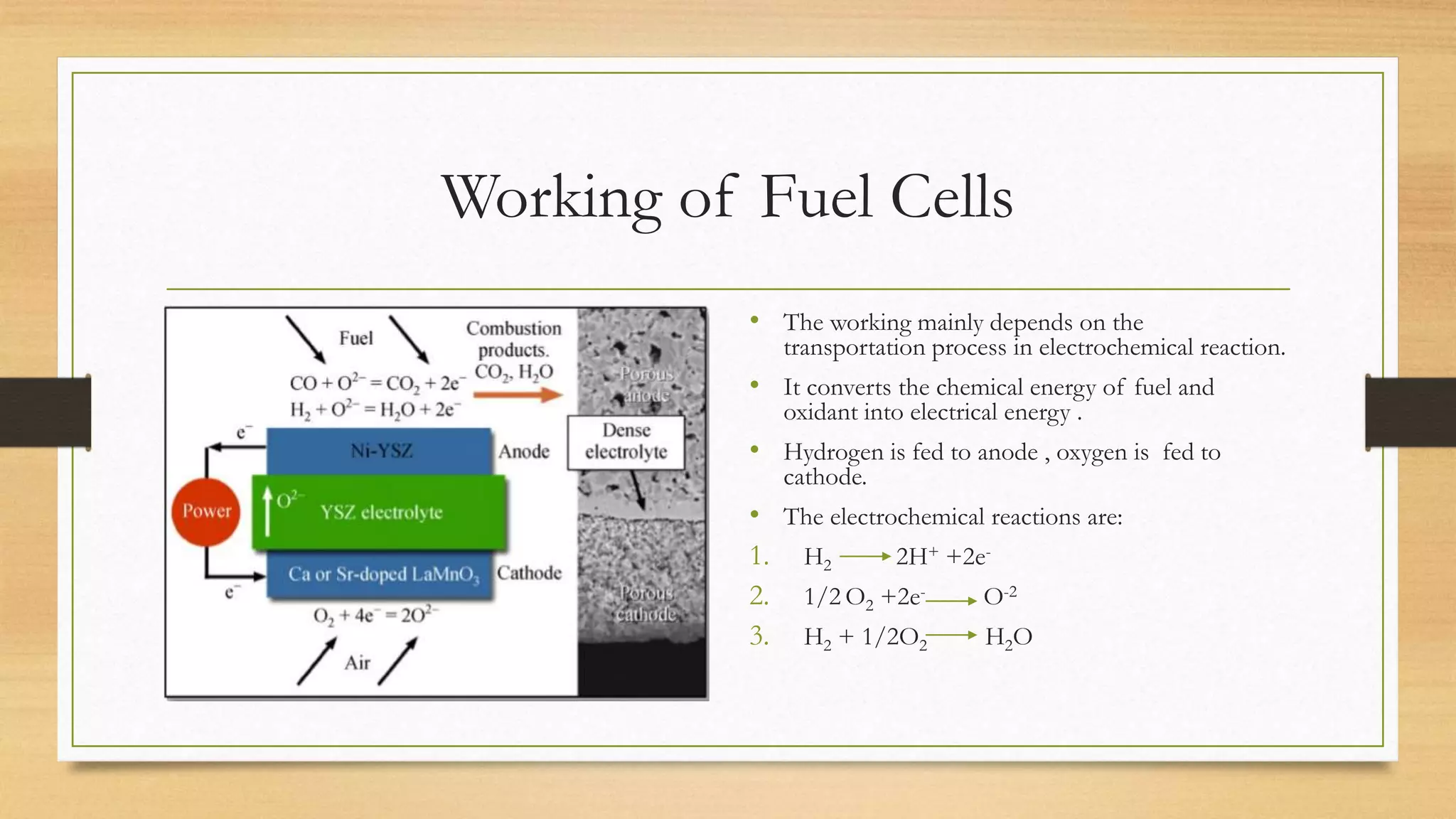
Solid Oxide Fuel Cells Pptx A solid oxide fuel cell (sofc) can be fed with several types of fuel, including hydrogen gas, carbon monoxide and methane, which are direct oxidation fuels within the cell. A solid oxide fuel cell (sofc) uses a hard ceramic electrolyte instead of a liquid and operates at temperatures up to 1,000 degrees c (about 1,800 degrees f). a mixture of zirconium oxide and calcium oxide form a crystal lattice, though other oxide combinations have also been used as electrolytes.
Comments are closed.