Section B How Much Do We Need To Worry About External Validity
External Validity Mike Denly Lecture 7 includes: section a: introduction, context, and framework; section b: how much do we need to worry about external validity; section c: how could we estimate the. In this paper we provide a formal, general exploration of the question of external validity and propose a simple and generally applicable method for evaluating the external validity of randomized controlled trials.

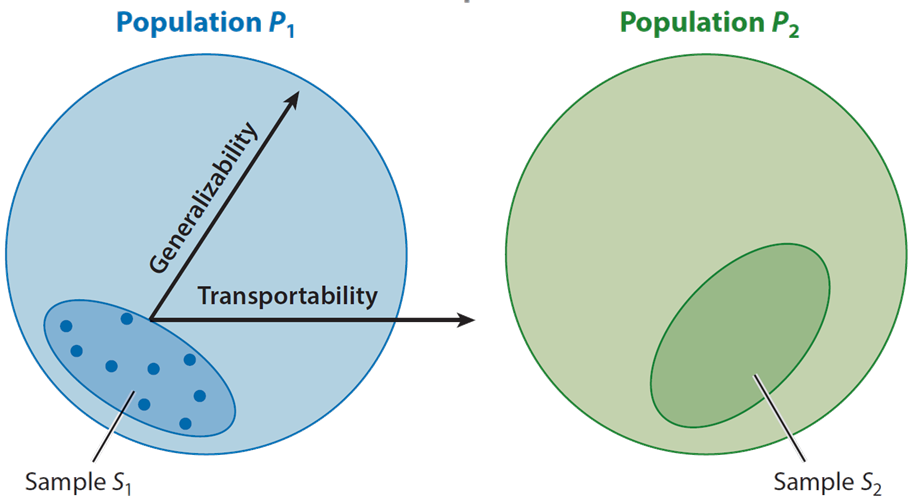
Examples Of External Validity In Research For any empirical causal study, one can decompose its validity into internal and external components. internal validity concerns whether the estimated effect is valid for the particular setting studied. external validity (ev), in contrast, looks beyond the sample studied. External validity and model validity of study results are important issues from a clinical point of view. from a methodological point of view, however, it appears that the concept of external validity and model validity is far more complex than it first seems. We first clarify the core conceptual dimensions of external validity and introduce a simple formalization that demonstrates why external validity matters so critically. External validity is an important parameter that needs to be considered for decision making in health research, but no widely accepted measurement tool for the assessment of external validity of randomized controlled trials (rcts) exists.

Pdf External Validity We Need To Do More We first clarify the core conceptual dimensions of external validity and introduce a simple formalization that demonstrates why external validity matters so critically. External validity is an important parameter that needs to be considered for decision making in health research, but no widely accepted measurement tool for the assessment of external validity of randomized controlled trials (rcts) exists. The central question in the use of research evidence in practice is how confident can we be that what is true for study participants (internal validity) is also true for other people (external validity). In the section formal framework for external validity, we developed a formal framework and discussed concerns for external validity. in this section, we outline our proposed approach to external validity, reserving details of our methods to the sections effect generalization and sign generalization. External validity, however, is typically harder to assess as it is difficult to know how a treatment effect may change in different populations. we review the measures of external validity, both those that focus solely on a single setting and those that compare across settings. We review, from a practical standpoint, the evolving literature on assessing external validity (ev) of estimated treatment effects. we review existing ev measures, and focus on methods that permit multiple datasets (hotz et al., 2005).

External Validity Threats Examples And Types Research Method The central question in the use of research evidence in practice is how confident can we be that what is true for study participants (internal validity) is also true for other people (external validity). In the section formal framework for external validity, we developed a formal framework and discussed concerns for external validity. in this section, we outline our proposed approach to external validity, reserving details of our methods to the sections effect generalization and sign generalization. External validity, however, is typically harder to assess as it is difficult to know how a treatment effect may change in different populations. we review the measures of external validity, both those that focus solely on a single setting and those that compare across settings. We review, from a practical standpoint, the evolving literature on assessing external validity (ev) of estimated treatment effects. we review existing ev measures, and focus on methods that permit multiple datasets (hotz et al., 2005).

External Validity Research Methods Knowledge Base External validity, however, is typically harder to assess as it is difficult to know how a treatment effect may change in different populations. we review the measures of external validity, both those that focus solely on a single setting and those that compare across settings. We review, from a practical standpoint, the evolving literature on assessing external validity (ev) of estimated treatment effects. we review existing ev measures, and focus on methods that permit multiple datasets (hotz et al., 2005).
Comments are closed.