Sars Cov 2 Live Virus Neutralization Assay
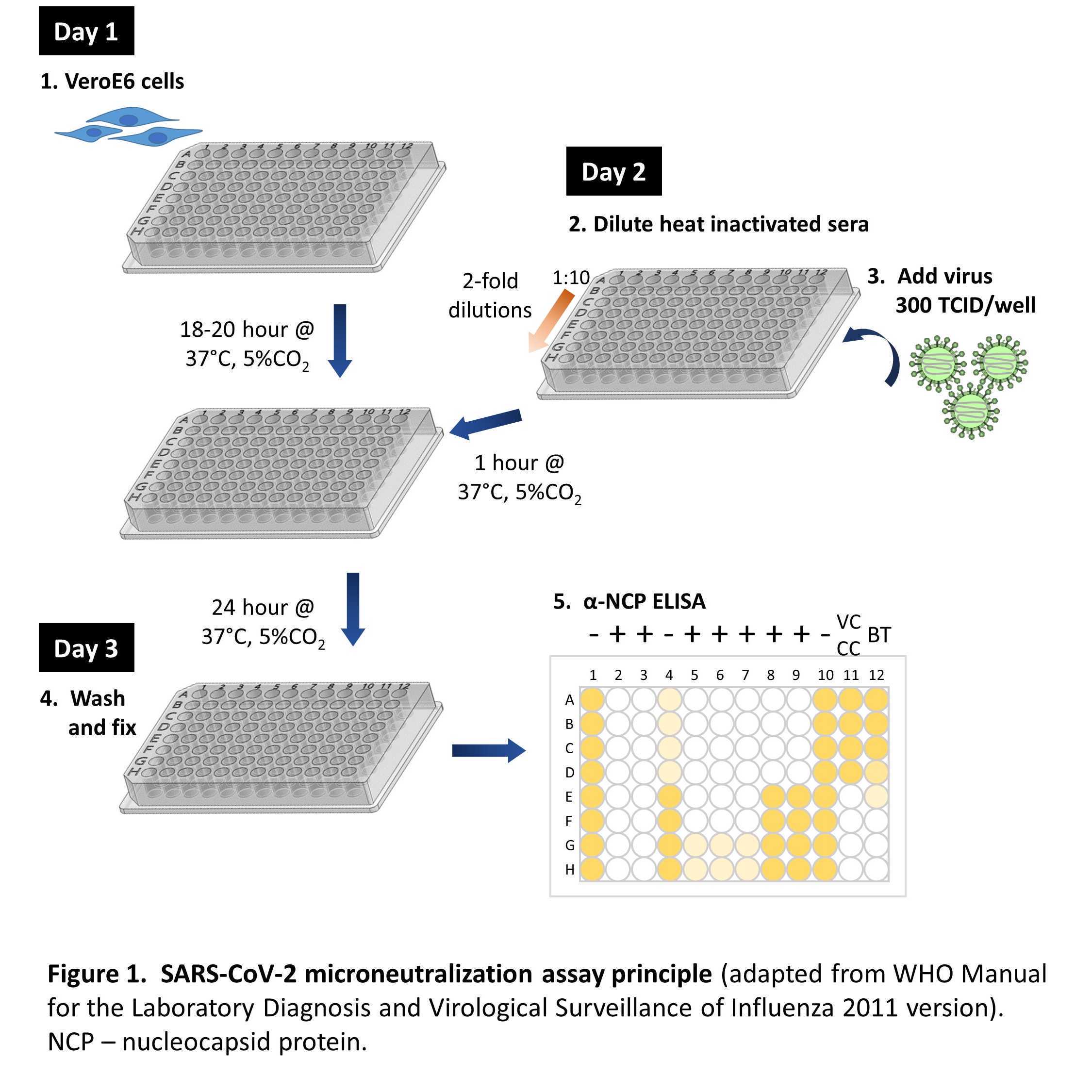
Sars Cov 2 Live Virus Neutralization Assay Here we describe the development, optimization and evaluation of a live virus microneutralization assay specific for severe acute respiratory syndrome coronavirus 2 (sars cov 2). in this assay, sars cov 2 clinical isolates are pre incubated with serial diluted antibody and added to vero e6 cells. Comparative analysis of neutralization assays performed using live sars cov 2 virus and pseudovirus to assess immunogenicity of a bivalent sars cov 2 protein vaccine in humans.

Pdf Quantifying Absolute Neutralization Titers Against Sars Cov 2 By Here we describe the development, optimization and evaluation of a live virus microneutralization assay specific for severe acute respiratory syndrome coronavirus 2 (sars cov 2). We have developed procedures for the standard prnt, microneutralization assay (mna) and pseudotyped virus neutralization assay (pna) for severe acute respiratory syndrome coronavirus. Here, we compared the performance of four virus neutralization tests to measure neutralizing antibodies in various sample types. Here, we present an integrated research–clinical platform for a live sars cov 2 neutralization assay, utilizing highly attenuated sars cov 2 (Δ3678 wa1 spike).
Neutralization Assay Neutralization Escape By Sars Cov 2 Omicron Here, we compared the performance of four virus neutralization tests to measure neutralizing antibodies in various sample types. Here, we present an integrated research–clinical platform for a live sars cov 2 neutralization assay, utilizing highly attenuated sars cov 2 (Δ3678 wa1 spike). In this study, therefore, we developed a faster live virus assay, which detects neutralizing antibodies through the early measure ment of antibody mediated intracellular virus reduction by sars cov 2 qrt pcr. Here we describe a pcr based micro‐neutralization assay that can be used to evaluate the viral neutralization titers of serum from sars cov 2 infected individuals. Why does the neutralizing antibody (nab) matter, and how does the cpass™ sars cov 2 nab detection kit by genscript works to detect the presence of the neutralizing antibodies. This narrative review examines the diverse neutralization assays utilized in clinical trials targeting sars cov 2. these assays, ranging from plaque reduction to pseudovirus and live virus assays, offer distinct advantages and considerations in terms of safety, sensitivity, and scalability.

Pdf Development Of A Rapid Live Sars Cov 2 Neutralization Assay Based In this study, therefore, we developed a faster live virus assay, which detects neutralizing antibodies through the early measure ment of antibody mediated intracellular virus reduction by sars cov 2 qrt pcr. Here we describe a pcr based micro‐neutralization assay that can be used to evaluate the viral neutralization titers of serum from sars cov 2 infected individuals. Why does the neutralizing antibody (nab) matter, and how does the cpass™ sars cov 2 nab detection kit by genscript works to detect the presence of the neutralizing antibodies. This narrative review examines the diverse neutralization assays utilized in clinical trials targeting sars cov 2. these assays, ranging from plaque reduction to pseudovirus and live virus assays, offer distinct advantages and considerations in terms of safety, sensitivity, and scalability.
Comments are closed.