Safety Risk Management Plan Rmp Pmda Atc E Learning

Raitek Rmp Risk Management Plan Pdf This video introduces factors consist of risk management plan (rmp) aiming to maintain the benefit of medicines such as pharmacovigilance and risk minimizati. 医薬品・医療機器・再生医療等製品の承認審査・安全対策・健康被害救済の3つの業務を行う組織。.

Tesda Rtc Vii Risk Management Plan 2022 Download Free Pdf Risk (safety) risk management plan (rmp) pmda atc e learning this video introduces factors consist of risk management plan (rmp) aiming to maintain the benefit of medicines such as pharmacovigilance and risk minimization activity. In this 2022, the indonesian fda, pharmaceuticals and medical devices agency (pmda, japan) and universitas indonesia (ui) by the support of japan asean integration fund (jaif) conduct a collaboration programme which is called asean japan pharmaceuticals training in risk management plan. This course introduces the risk management plan (rmp) and the related requirements, structure, content, and activities. terms such as "safety concern" and "important risk" will be explained according to the gvp v rev. 2 guidance and compared to definitions and use in other pharmacovigilance documents. The primary aim and focus of the rmp remains that of appropriate risk management planning throughout a medicinal product’s life cycle. it is designed to optimise the benefit risk balance over time.
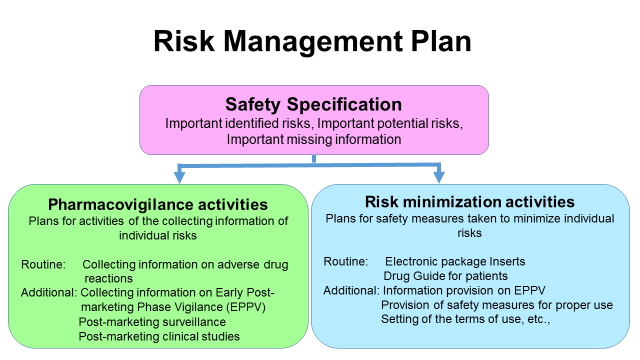
Risk Management Plan Rmp Pharmaceuticals And Medical Devices Agency This course introduces the risk management plan (rmp) and the related requirements, structure, content, and activities. terms such as "safety concern" and "important risk" will be explained according to the gvp v rev. 2 guidance and compared to definitions and use in other pharmacovigilance documents. The primary aim and focus of the rmp remains that of appropriate risk management planning throughout a medicinal product’s life cycle. it is designed to optimise the benefit risk balance over time. This course will be helpful to those who have icsr, aggregate reporting or signal management experience in pharmacovigilance domain and for those who wants to learn and excel their career in pharmacovigilance risk management plan (rmp). This risk management plan (rmp) facilitates proactive risk management and serves as a guide for developing individual plans. expert guidance is available for creating a customized risk management plan with a downloadable template. The pharmaceuticals and medical devices agency (pmda) is pleased to announce the “pmda atc pharmacovigilance webinar 2024” for officials from overseas regulatory authorities who are engaged in pharmacovigilance activities. The rmp includes a general overview, safety specifications, a pharmacovigilance plan, and risk minimization measures tailored to specific concerns. download as a pdf, pptx or view online for free.
Comments are closed.