S Block Elements S Block Elements In Periodic Table

Modern Periodic Table With S Block Elements Stock Illustration In this article, we will learn about s block elements, their various properties, and others in detail. Learn about the s block elements on the periodic table. get a list of the elements and learn about their common properties.

Periodic Table D Block Elements Names Cabinets Matttroy The "s block" in the periodic table refers to the two groups of elements located in the leftmost part of the periodic table: group 1 and group 2. these elements are characterized by their outermost electrons occupying the s orbital of their respective electron shells. Each block is named after its characteristic orbital: s block, p block, d block, f block and g block. the block names (s, p, d, and f) are derived from the spectroscopic notation for the value of an electron's azimuthal quantum number: sharp (0), principal (1), diffuse (2), and fundamental (3). S block elements are those whose outermost electrons occupy an s orbital, comprising group 1 (alkali metals) and group 2 (alkaline earth metals). some sources also consider hydrogen and helium —though helium is a noble gas, it has its electron in the s orbital. The s block elements of the periodic table are those elements in which the last electron enters the outermost s subshell. as the s subshell can accommodate only two electrons, the first two groups belong to the s block of the periodic table.

S Block Elements From The Modern Periodic Table Stock Illustration S block elements are those whose outermost electrons occupy an s orbital, comprising group 1 (alkali metals) and group 2 (alkaline earth metals). some sources also consider hydrogen and helium —though helium is a noble gas, it has its electron in the s orbital. The s block elements of the periodic table are those elements in which the last electron enters the outermost s subshell. as the s subshell can accommodate only two electrons, the first two groups belong to the s block of the periodic table. What are the s block elements of the periodic table. check out their list and learn their characteristics. The s block in the periodic table of elements occupies the alkali metals and alkaline earth metals, also known as groups 1 and 2. helium is also part of the s block. S block elements are elements located in groups 1 and 2 of the periodic table, including the alkali metals and alkaline earth metals. they have their valence electrons in the 's' orbital, which gives them characteristic chemical properties. Learn how to identify s, p, d, and f block elements, and see examples that walk through sample problems step by step for you to improve your chemistry knowledge and skills.
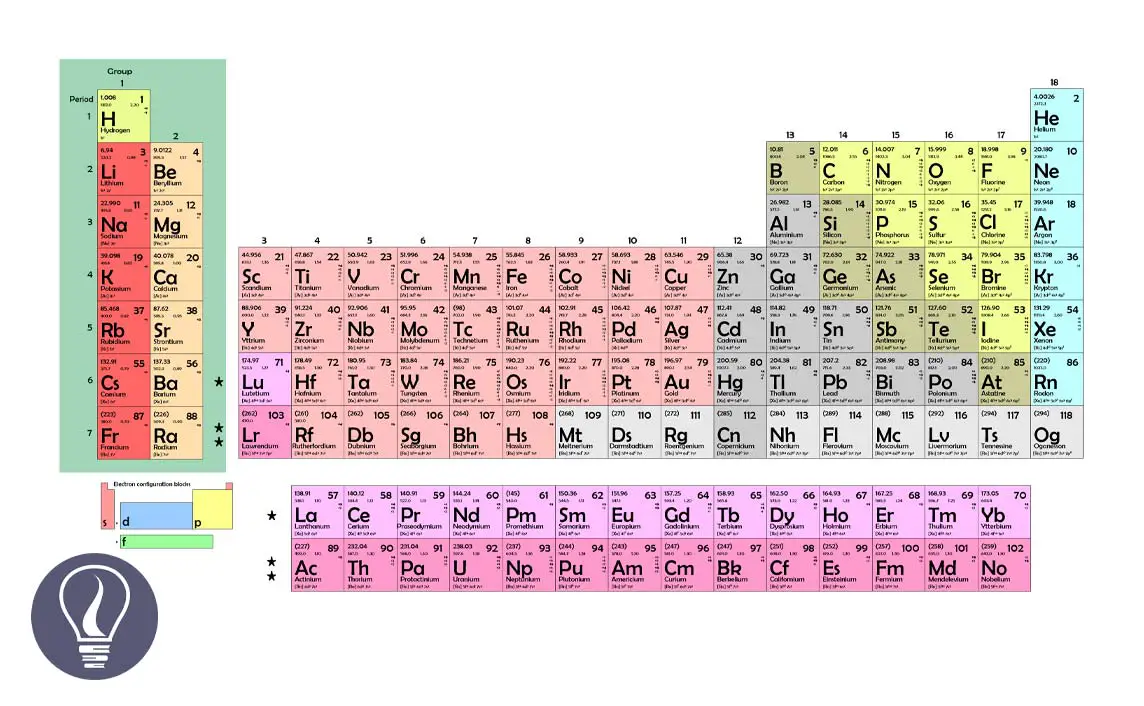
S Block Elements In The Periodic Table Learnbin What are the s block elements of the periodic table. check out their list and learn their characteristics. The s block in the periodic table of elements occupies the alkali metals and alkaline earth metals, also known as groups 1 and 2. helium is also part of the s block. S block elements are elements located in groups 1 and 2 of the periodic table, including the alkali metals and alkaline earth metals. they have their valence electrons in the 's' orbital, which gives them characteristic chemical properties. Learn how to identify s, p, d, and f block elements, and see examples that walk through sample problems step by step for you to improve your chemistry knowledge and skills.

S Block Elements Group 1 And Group 2 Elements Periodic Table Notes S block elements are elements located in groups 1 and 2 of the periodic table, including the alkali metals and alkaline earth metals. they have their valence electrons in the 's' orbital, which gives them characteristic chemical properties. Learn how to identify s, p, d, and f block elements, and see examples that walk through sample problems step by step for you to improve your chemistry knowledge and skills.
Comments are closed.