S Block Elements Pdf Solvation Ion

S Block Elements Group 1 Pdf Pdf Ion Ionic Bonding The document discusses the characteristic properties of the s block elements. it describes their metallic character, low electronegativity, formation of basic oxides and hydroxides, and fixed oxidation states. These metals form diapositive ions (m 2). these diapositive ions have noble gas configuration and their compounds are colourless, because all the electrons are paired.

S Block Elements Pdf Sodium Hydroxide Sodium The s block elements of the periodic table are those in which the last electron enters the outermost s orbital. as the s orbital can accommodate only two electrons, two groups (1 & 2) belong to the s block of the periodic table. Alkali metals are soft with low melting and boiling points. this is due to weak metallic bond. solution of alkali metals in liquid ammonia is highly conductive and deep blue in colour. Sodium ions are involved in the transmission of nerve signals, in regulating the flow of water across the cell membranes, and in transporting sugars and amino acids into the cells. Summary to s orbital are s block elements. alkali metals: the elements of group 1 whose hydroxide are strong alkali. alkaline earth metal: the elements of group 2, and their oxides and hydroxides are alkaline in nature and their oxides are found in the earth’s crust.

S Block Elements 2021 22 Pdf Ion Hydroxide Sodium ions are involved in the transmission of nerve signals, in regulating the flow of water across the cell membranes, and in transporting sugars and amino acids into the cells. Summary to s orbital are s block elements. alkali metals: the elements of group 1 whose hydroxide are strong alkali. alkaline earth metal: the elements of group 2, and their oxides and hydroxides are alkaline in nature and their oxides are found in the earth’s crust. Class 11 chemistry book chapter 3. the s – block elements in english download in pdf. books for class 6 to 12 all subjects chapter wise in hindi and english medium. Beryllium dissolves in alkalies to give beryllate ion [ be(oh)4]2 while aluminium dissolves to give [ al(oh)6]3 becl2 like al2cl6 has a bridged polymeric structure similar solubility are observed in halides of both beryllium and aluminium. Ionic hydrides: they are formed when hydrogen molecule reacts with highly electropositive s block elements (alkali metals and alkaline earth metals). in solid state, the ionic hydrides are crystalline, non conducting and non volatile. Larger hydration enthalpy than that of the corresponding alkali metal ions. this is why compounds of alkaline earth metals are more extensively hydrated than those of alkali metals.
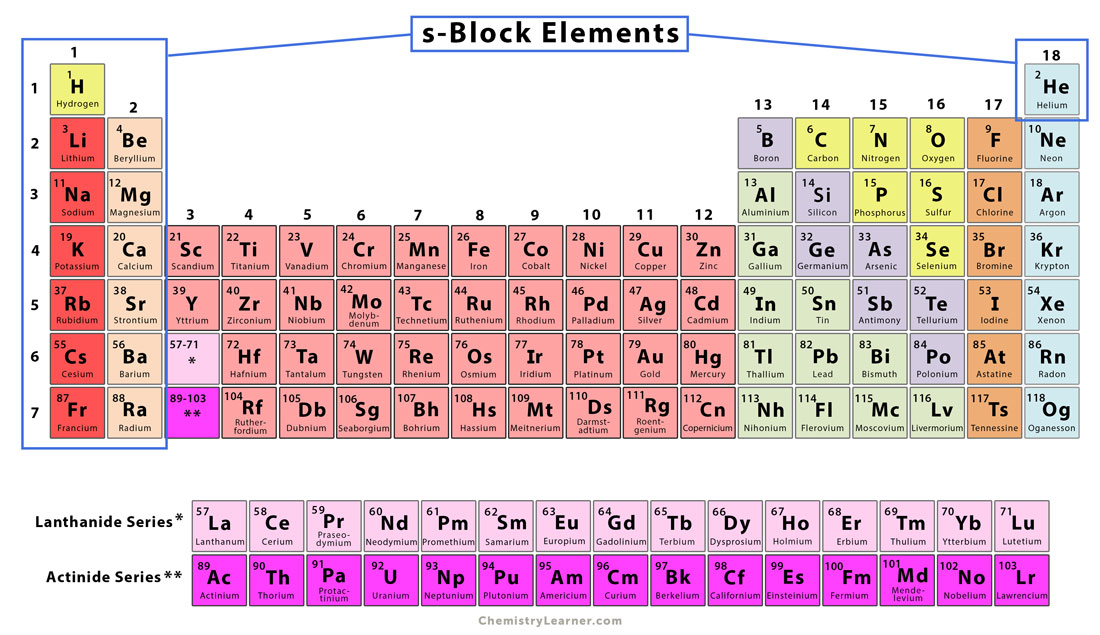
S Block Elements Definition And Characteristics Class 11 chemistry book chapter 3. the s – block elements in english download in pdf. books for class 6 to 12 all subjects chapter wise in hindi and english medium. Beryllium dissolves in alkalies to give beryllate ion [ be(oh)4]2 while aluminium dissolves to give [ al(oh)6]3 becl2 like al2cl6 has a bridged polymeric structure similar solubility are observed in halides of both beryllium and aluminium. Ionic hydrides: they are formed when hydrogen molecule reacts with highly electropositive s block elements (alkali metals and alkaline earth metals). in solid state, the ionic hydrides are crystalline, non conducting and non volatile. Larger hydration enthalpy than that of the corresponding alkali metal ions. this is why compounds of alkaline earth metals are more extensively hydrated than those of alkali metals.
Comments are closed.