S Block Elements Pdf Periodic Table Lithium

S Block Elements Download Free Pdf Periodic Table Lithium S block elements free download as pdf file (.pdf), text file (.txt) or read online for free. s block elements are those whose outermost electrons are in the s orbital. As the s orbital can accommodate only two electrons, two groups (1 & 2) belong to the s block of the periodic table. group 1 of the periodic table consists of the elements: lithium, sodium, potassium, rubidium, caesium and francium. they are collectively known as the alkali metals.

Printable Periodic Table Lithium Free Download And Print For You The long form of the periodic table has been conveniently divided into four blocks i.e., s, p, d and f blocks, depending upon the filling of a particular shell. Results in the increased covalent character of lithium compounds. li is much harder. m.p. and b.p. are higher than other alkali metals. licl is deliquescent and crystallises as a hydrate, licl.2h2o. Y the lithium, being the first member of alkali group, exhibits the characteristic properties of alkali metals but it differs at the same time in many respects from them. Exceptionally small size of its atom and ion. greater polarizing power of lithium ion. as compared to other alkali metals, lithium is harder and its melting point and boiling point are higher. among all the alkali metals lithium is least reactive but the strongest reducing agent.
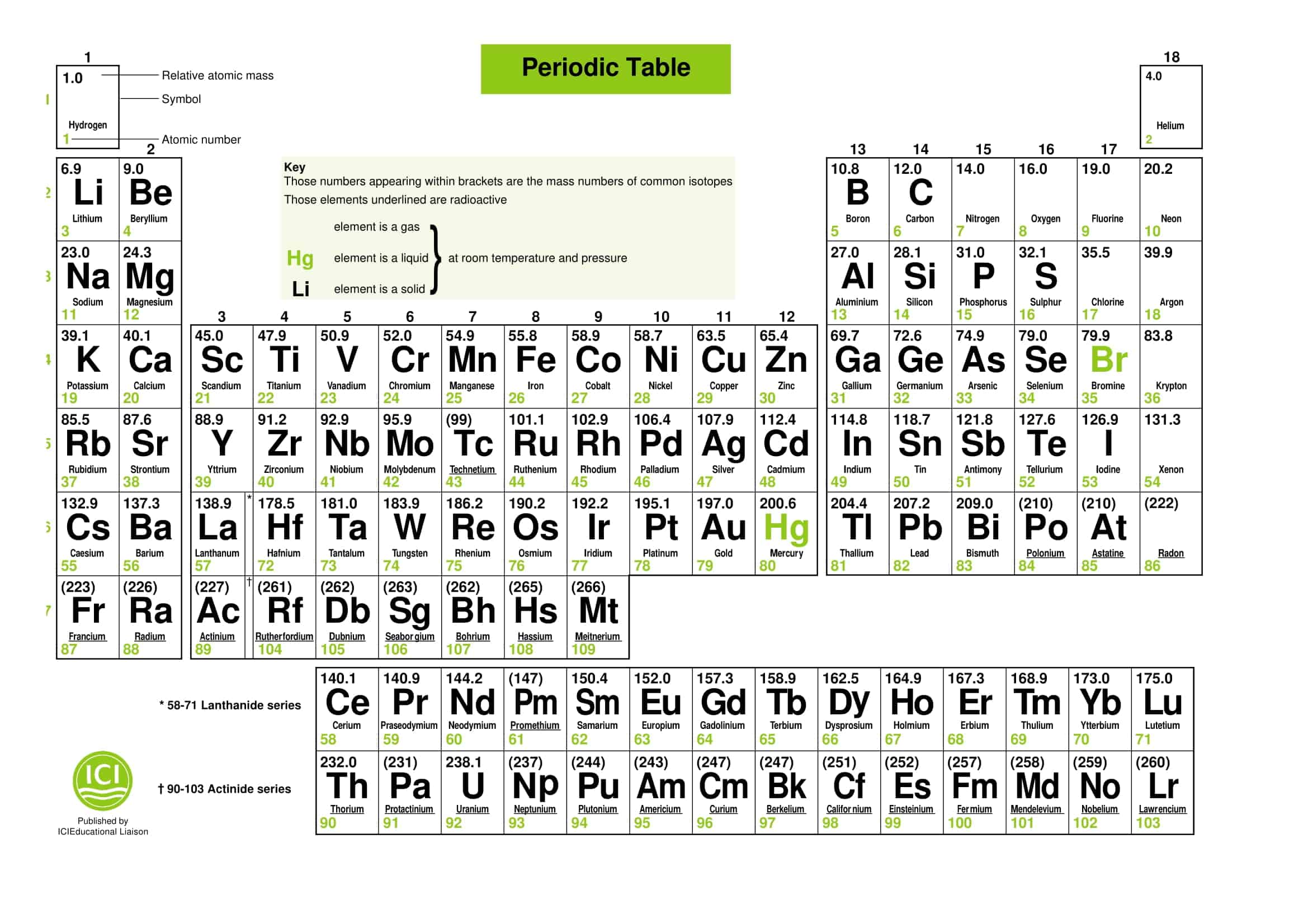
Periodic Table Of Elements Printable Pdf Infoupdate Org Y the lithium, being the first member of alkali group, exhibits the characteristic properties of alkali metals but it differs at the same time in many respects from them. Exceptionally small size of its atom and ion. greater polarizing power of lithium ion. as compared to other alkali metals, lithium is harder and its melting point and boiling point are higher. among all the alkali metals lithium is least reactive but the strongest reducing agent. As the s orbital can accommodate only two electrons, two groups (1 & 2) belong to the s block of the periodic table. group 1 of the periodic table consists of the elements: lithium, sodium, potassium, rubidium, caesium and francium. they are collectively known as the alkali metals. Lithium forms monoxide, sodium forms peroxide, the other metals form superoxides. the superoxide o2 – ion is stable only in the presence of large cations such as k , rb , cs due to stabilisation of larger anions by larger cations through lattice energy. Learn about the s block elements on the periodic table. get a list of the elements and learn about their common properties. In this article, we will learn about s block elements, their various properties, and others in detail.
Comments are closed.