Risk Management In The Design Of Medical Device Software Systems Pdf

Risk Management In The Design Of Medical Device Software Systems Pdf Aspects of change implementation contribute to the accurate depiction of a device and or its risk profile. medical device software can pose risks that are distinct and unique, such as those that relate to the infor. The purpose of this paper is to research similarities and differences between iso 14971 risk management process and agile principles.

Understanding Integrated Risk Management For Medical Devices Jama Software in medical devices module 3 free download as pdf file (.pdf), text file (.txt) or view presentation slides online. this document discusses software risk management in medical devices. it begins with an introduction to risk management concepts and murphy's law. Tr 80002 1:2009 (iec tr 2009) provides guidance on the application of iso 14971 (iso 2012) to medical device software. two other examples of guidance documents are, do it by design (fda 1996), introducing human factors in medical devices development and medical device use. In health care it is essential to manage risk related to software due to its increased use in medical devices and other computer systems. this paper presents some of the experiences gained from an ongoing case study at a large hospital in sweden. The development of medical device software is strictly regulated by competent authorities. in addition to producing significant medical benefits, the medical device software can be a potential source of serious safety hazard to patients or healthcare professionals.
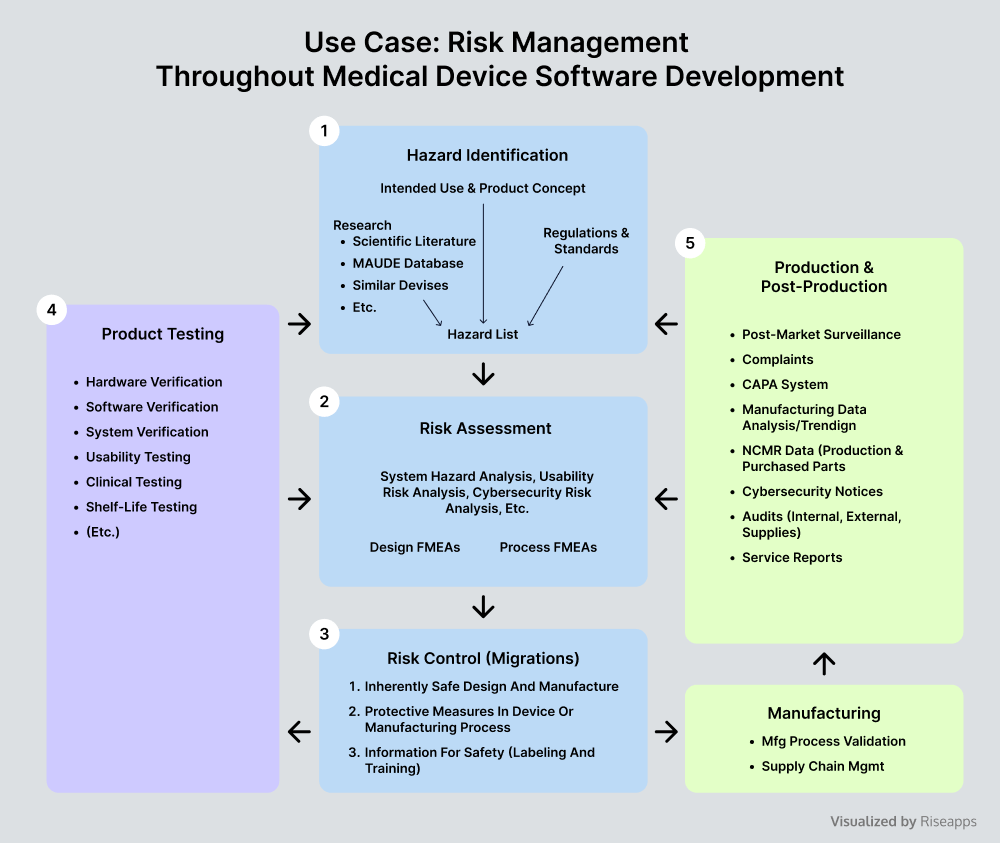
Medical Device Software Development Risks Vs Potentials In health care it is essential to manage risk related to software due to its increased use in medical devices and other computer systems. this paper presents some of the experiences gained from an ongoing case study at a large hospital in sweden. The development of medical device software is strictly regulated by competent authorities. in addition to producing significant medical benefits, the medical device software can be a potential source of serious safety hazard to patients or healthcare professionals. Software failures in medical devices can lead to catastrophic situations. therefore, it is crucial to handle software related risks when developing medical devices, and there is a need for further analysis of how this type of risk management should be conducted. Software failures in medical devices can lead to catastrophic situations. therefore is it crucial to handle software related risks when developing medical devices. this paper presents the experiences gained from an ongoing case study with a medical device development organisation. It is widely acknowledged as the primary risk management standard for medical devices, including software as a medical device (thereby including security risks) and in vitro diagnostic devices. Report. ultimately, such a system poses risks to end users, stakeholders, and the company relying on the risk management process and results to bring a product safely to market.

Medical Device Risk Management Expert Guide Scilife Software failures in medical devices can lead to catastrophic situations. therefore, it is crucial to handle software related risks when developing medical devices, and there is a need for further analysis of how this type of risk management should be conducted. Software failures in medical devices can lead to catastrophic situations. therefore is it crucial to handle software related risks when developing medical devices. this paper presents the experiences gained from an ongoing case study with a medical device development organisation. It is widely acknowledged as the primary risk management standard for medical devices, including software as a medical device (thereby including security risks) and in vitro diagnostic devices. Report. ultimately, such a system poses risks to end users, stakeholders, and the company relying on the risk management process and results to bring a product safely to market.

Software In Medical Devices Module 3 Pdf Risk Management Hazards It is widely acknowledged as the primary risk management standard for medical devices, including software as a medical device (thereby including security risks) and in vitro diagnostic devices. Report. ultimately, such a system poses risks to end users, stakeholders, and the company relying on the risk management process and results to bring a product safely to market.
Comments are closed.