Rheology Natoliscientific Tabletdevelopment Formulation

Formulation And Optimization Of Immediate Release Tablet Of An Anti Natoli scientific has established new and unique procedures to predict the formulation’s tendency for sticking along with recommendations for the right steel type to reduce its likelihood. Our designed ‘natoli in house’ testing approach coupled with ft4 rheometer along with dynamic compaction profiling will help you to decode the influence of material properties and tooling composition on tablet sticking, picking, and lamination.
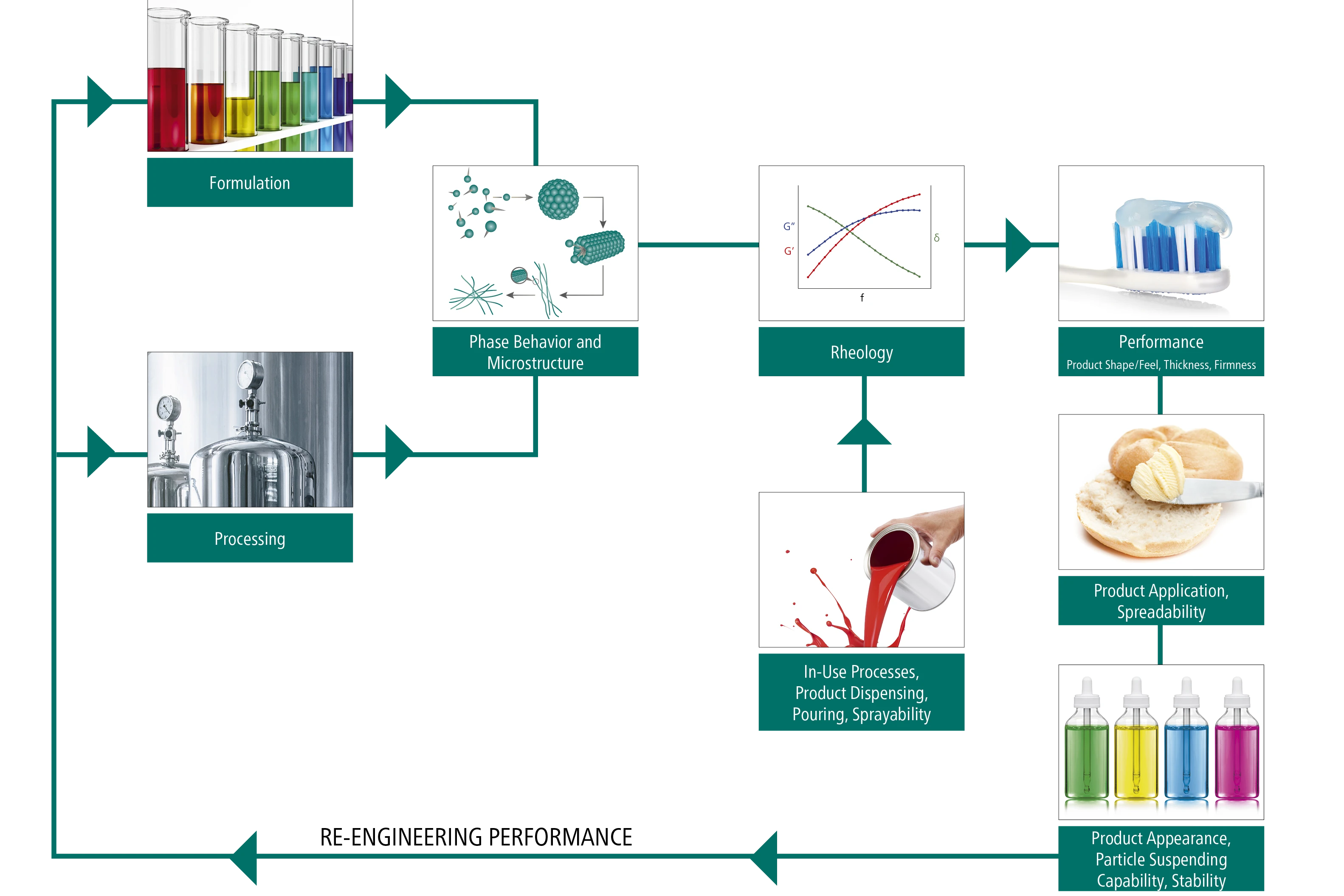
Using Rheology For Faster Smarter Material Formulation Netzsch The study concludes that powder rheology testing provides valuable early insight into sticking risks, allowing manufacturers to refine formulation and tooling choices before scale up. 📈🔍the physical properties of powders, such as how they flow, pack, and compress, can be measured by powder rheology. Download the brochure to learn more on natoli’s formulation development solutions. we offer comprehensive services to improve processes so you can decrease development times and avoid scale up challenges and common manufacturing issues, such as sticking, picking, capping, and lamination. Natoli scientific experts can recommend an efficient and effective tablet design for your product based on your formulation’s characteristics and your manufacturing requirements and restrictions.

Understanding Rheology In Pharmaceutical Formulation Galaxy Ai Download the brochure to learn more on natoli’s formulation development solutions. we offer comprehensive services to improve processes so you can decrease development times and avoid scale up challenges and common manufacturing issues, such as sticking, picking, capping, and lamination. Natoli scientific experts can recommend an efficient and effective tablet design for your product based on your formulation’s characteristics and your manufacturing requirements and restrictions. Tablet sticking is an outcome of improper blend properties and poor tooling health of the tablet press. natoli scientific has established new and unique procedures to predict the formulation’s tendency for sticking along with recommendations for the right steel type to reduce its likelihood. Natoli scientific, in collaboration with tablitz, conducted a case study to evaluate a customer’s formulation, adhering to usp <1062> principles for tabletability, compressibility, and compactibility. In this study, systematic investigations into the formulation development of tablets containing polymeric and other excipients are performed by water uptake and swelling analysis. the impact of tablet composition and porosity as well as ph of the test medium are investigated. The document discusses the importance of performing detailed formulation development and compaction studies before final regulatory submission. this helps ensure formulation performance and avoids potential production problems.

Rheology For Material Formulation Malvern Panalytical Tablet sticking is an outcome of improper blend properties and poor tooling health of the tablet press. natoli scientific has established new and unique procedures to predict the formulation’s tendency for sticking along with recommendations for the right steel type to reduce its likelihood. Natoli scientific, in collaboration with tablitz, conducted a case study to evaluate a customer’s formulation, adhering to usp <1062> principles for tabletability, compressibility, and compactibility. In this study, systematic investigations into the formulation development of tablets containing polymeric and other excipients are performed by water uptake and swelling analysis. the impact of tablet composition and porosity as well as ph of the test medium are investigated. The document discusses the importance of performing detailed formulation development and compaction studies before final regulatory submission. this helps ensure formulation performance and avoids potential production problems.
Comments are closed.