Research Methodologies Introduction To Validation And Verification

Introduction To Pharmaceutical Validation Pdf Verification And In preparing this guidance, the working group has aimed at the right balance between giving a solid theoretical background for method validation studies and providing practical guidelines on how to plan, perform and evaluate such studies in the laboratories. R the derivation of theorems that clarify the conceptual underpinnings of v&v. by providing a formal foundation, this work addresses ambiguities in traditional v&v practices, offering a structured frame.

Validation Vs Verification What S The Difference Verification can be in development, scale up, or production. often an internal process. validation is a quality assurance process establishing evidence that provides a high degree of assurance that a product, service, or system accomplishes its intended requirements. This course provides an introduction to validation and verification of analytical methods. the common elements of a validation verification plan and a general approach to performing a validation or verification are presented. Validation is an applied approach to verify that a method is suitable to function as a quality control tool. the objective of any analytical measurement is to obtain consistent, reliable and. It establishes the foundation for effective data collection, analysis, and interpreta tion, ensuring that research findings are reliable. a well designed research method ology also enhances the reproducibility of research, allowing other researchers to replicate and validate the study’s result.
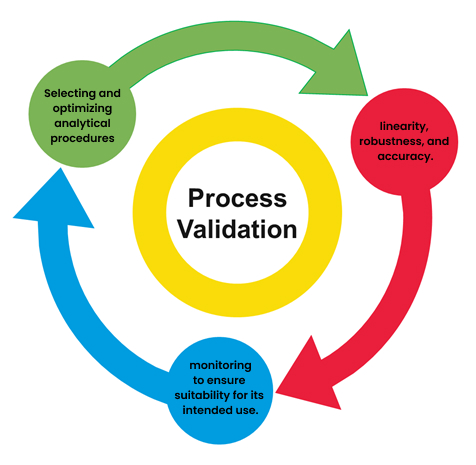
Method Validation Verification Pls Analytical Validation is an applied approach to verify that a method is suitable to function as a quality control tool. the objective of any analytical measurement is to obtain consistent, reliable and. It establishes the foundation for effective data collection, analysis, and interpreta tion, ensuring that research findings are reliable. a well designed research method ology also enhances the reproducibility of research, allowing other researchers to replicate and validate the study’s result. Basic validation and verification is the plain language, common sense guide to the studies and statistics that govern the evaluation, validation and verification of a laboratory method. Stematic evaluation of analytical methods, known as method validation. according to the international conference on harmonisation (ich) guidelines, method validation confirms that an analytical procedure is suitab. This comprehensive guide explores the principles of analytical method development and validation, covering key definitions, core validation parameters, regulatory frameworks, and practical considerations for laboratory managers. References and additional reading material listed at the end of this document may provide useful and further guidance on the verification and validation of methods.
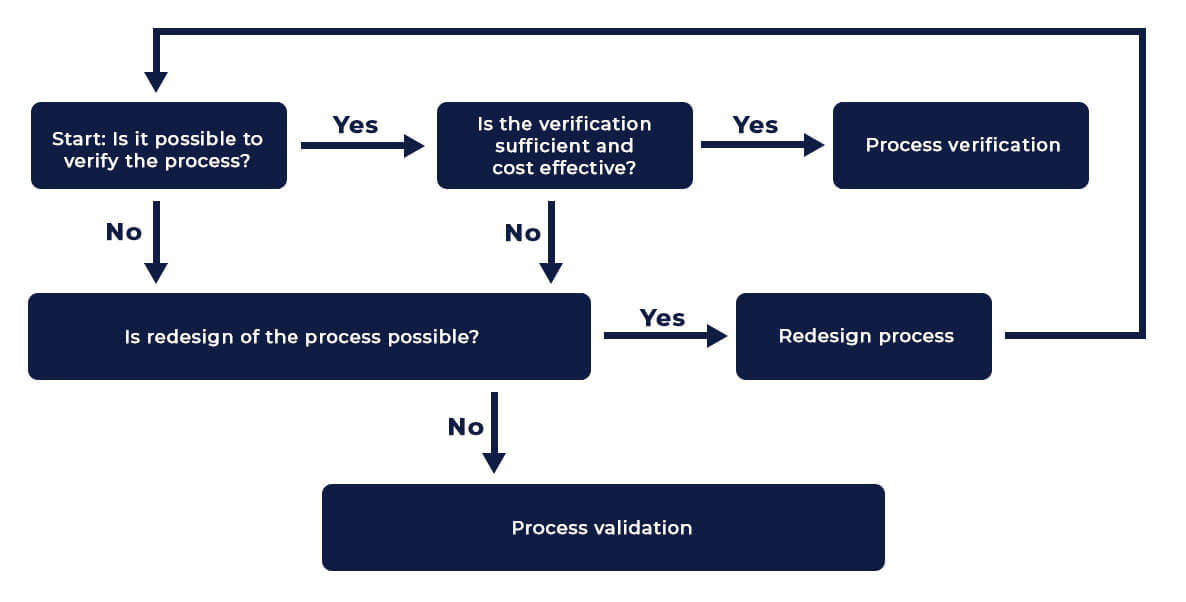
Verification Vs Validation Basic validation and verification is the plain language, common sense guide to the studies and statistics that govern the evaluation, validation and verification of a laboratory method. Stematic evaluation of analytical methods, known as method validation. according to the international conference on harmonisation (ich) guidelines, method validation confirms that an analytical procedure is suitab. This comprehensive guide explores the principles of analytical method development and validation, covering key definitions, core validation parameters, regulatory frameworks, and practical considerations for laboratory managers. References and additional reading material listed at the end of this document may provide useful and further guidance on the verification and validation of methods.
Comments are closed.