Research Methodologies Data Validation And Verification

Data Validation Vs Data Verification What S The Differences It involves cross checking, validating, and correcting data to ensure that it aligns with the research objectives and methods, minimizing errors and bias. this article explores data verification in the context of research, detailing its process, types, and examples. This paper explores key data validation techniques, including range checks, type checks, code validation, uniqueness checks, and consistency checks. it also distinguishes between automated.
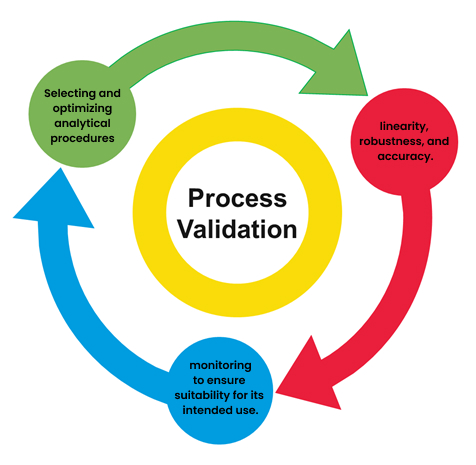
Method Validation Verification Pls Analytical Research data is loosely defined as information collected, observed, or created for purposes of analysis to produce original research. this guide provides resources for managing your research data no matter the discipline. Verification and validation (v&v) sit at the core of these dual aspirations. yet, despite their foundational role, v&v remain unevenly articulated and inconsistently practiced within design research methodology. The importance of validation is strengthened, more instructions are issued for the validation of analytical methods (ec 657 2002) as well as the performance criteria (ec 333 2007, ec 401 2006, etc.). This checklist (data source verification, accuracy, unit consistency, etc.) helps researchers ensure data integrity, spot errors, and maintain compliance crucial for reliable results and reproducible science.
Validation Vs Verification What S The Difference The importance of validation is strengthened, more instructions are issued for the validation of analytical methods (ec 657 2002) as well as the performance criteria (ec 333 2007, ec 401 2006, etc.). This checklist (data source verification, accuracy, unit consistency, etc.) helps researchers ensure data integrity, spot errors, and maintain compliance crucial for reliable results and reproducible science. Master 10 essential data validation techniques to build reliable pipelines. learn ai powered solutions, implementation steps & avoid common pitfalls. Through these methods, automated data validation acts as the unsung hero, a bulwark against the chaos of erroneous data, ensuring that market research and analysis stand on a foundation of veracity. The absence of a well defined data quality definition and method to measure error undermines the reliability of data quality assessment. this review aimed to assess the variability of source data verification (sdv) auditing methods to monitor data quality in a clinical research setting. I. purpose requirements to perform a validation or verification study. validations must be performed for all non st ndard and laboratory developed methods. verifications must be performed for all unmodified standard method.

Validation Vs Verification What S The Difference Master 10 essential data validation techniques to build reliable pipelines. learn ai powered solutions, implementation steps & avoid common pitfalls. Through these methods, automated data validation acts as the unsung hero, a bulwark against the chaos of erroneous data, ensuring that market research and analysis stand on a foundation of veracity. The absence of a well defined data quality definition and method to measure error undermines the reliability of data quality assessment. this review aimed to assess the variability of source data verification (sdv) auditing methods to monitor data quality in a clinical research setting. I. purpose requirements to perform a validation or verification study. validations must be performed for all non st ndard and laboratory developed methods. verifications must be performed for all unmodified standard method.

Verification Vs Validation Board Infinity The absence of a well defined data quality definition and method to measure error undermines the reliability of data quality assessment. this review aimed to assess the variability of source data verification (sdv) auditing methods to monitor data quality in a clinical research setting. I. purpose requirements to perform a validation or verification study. validations must be performed for all non st ndard and laboratory developed methods. verifications must be performed for all unmodified standard method.
Comments are closed.