Redox Reactions Definition Examples Types Balancing

Balancing Redox Reactions Examples Pdf Redox Chemical Reactions Learn about redox reactions, their definition, examples, types, and applications in daily life. understand oxidation, reduction, and balancing redox reactions with clarity. Ans. balancing redox reactions is different from balancing other reactions. a half reaction is used in a redox reaction where the number of atoms and the amount of charge must be balanced.

Redox Reactions Examples Types Applications Balancing Pdf Oxidation reduction reactions, or redox reactions, are reactions in which one reactant is oxidized and one reactant is reduced simultaneously. this module demonstrates how to balance various redox equations. In this comprehensive study material, prof. neeraj anand of anand classes explains the concept of redox reactions with clear definitions, solved examples, different types of redox processes, and easy to follow steps for balancing redox equations. A redox (reduction oxidation) reaction involves the transfer of electrons between two substances. this type of reaction is fundamental in chemistry and is central to many biological and industrial processes. The most common redox reactions are combination reactions, decomposition reactions, combustion reactions, and displacement reactions.

1 1 Balancing Redox Reactions Pdf Chemical Reactions Redox A redox (reduction oxidation) reaction involves the transfer of electrons between two substances. this type of reaction is fundamental in chemistry and is central to many biological and industrial processes. The most common redox reactions are combination reactions, decomposition reactions, combustion reactions, and displacement reactions. We can balance redox reactions in two ways ion electron method and the oxidation number method. the ion electron method is based on the loss and gain of electrons during the redox process. Master the process of balancing redox reactions using oxidation numbers and the half reaction method. learn how electrons are transferred and conserved in electrochemical reactions. Learn what a redox reaction is and how to identify and balance redox reactions. explore oxidation, reduction, and oxidation numbers. Consider the following two reactions in which the removal of oxygen and addition of hydrogen take place respectively. these reactions are called redox reactions. cuo c → cu co (removal of oxygen from cupric oxide) s h2 → h2s (addition of hydrogen to sulphur).

Balancing Redox Reactions Facts Summary Definition Chemistry We can balance redox reactions in two ways ion electron method and the oxidation number method. the ion electron method is based on the loss and gain of electrons during the redox process. Master the process of balancing redox reactions using oxidation numbers and the half reaction method. learn how electrons are transferred and conserved in electrochemical reactions. Learn what a redox reaction is and how to identify and balance redox reactions. explore oxidation, reduction, and oxidation numbers. Consider the following two reactions in which the removal of oxygen and addition of hydrogen take place respectively. these reactions are called redox reactions. cuo c → cu co (removal of oxygen from cupric oxide) s h2 → h2s (addition of hydrogen to sulphur).
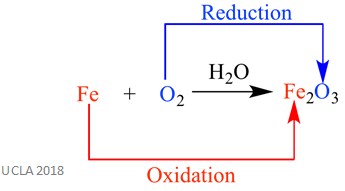
Balancing Redox Reactions Facts Summary Definition Chemistry Learn what a redox reaction is and how to identify and balance redox reactions. explore oxidation, reduction, and oxidation numbers. Consider the following two reactions in which the removal of oxygen and addition of hydrogen take place respectively. these reactions are called redox reactions. cuo c → cu co (removal of oxygen from cupric oxide) s h2 → h2s (addition of hydrogen to sulphur).
Comments are closed.