Real World Evidence Analytics Quantify Research

Real World Evidence Analytics Quantify Research Quantify is the leading provider of nordic real world evidence (rwe) with vast documented experience in high impact rwd studies. the quantify team possess expertise in data access, analysis, and interpretation with an unparalleled understanding of nordic data in denmark, finland, norway, and sweden. Founded in 2011, quantify research serves nineteen of the top twenty global pharmaceutical companies through three core practice areas: real world evidence and analytics (rwea), modelling, access & strategy (mas), and evidence review & synthesis (ers).
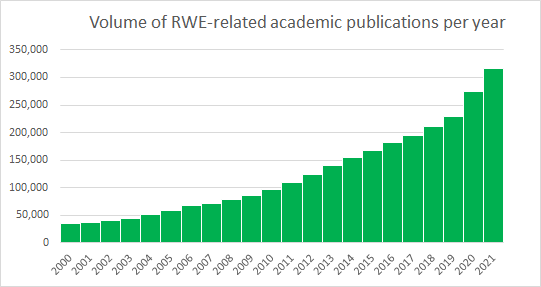
What Is Real World Evidence Quantify Research Real world evidence analysis represents a transformative frontier in healthcare research. by systematically harnessing data from routine clinical practice, rwe offers the promise of faster, more inclusive and more practical evidence on the safety and effectiveness of medical interventions. ️ our three specialized units modelling, access & strategy (mas), real world evidence & analytics (rwea), and evidence review & synthesis (ers) – work seamlessly to deliver comprehensive. The understanding of real world evidence has gained traction in recent years, with an increasing number of possible uses, from healthcare product development and approval to the final clinical decision and guidance of public policies. Adoption of rwd e in research and development (r&d) continues to accelerate, and there has been an expansion of rwd e use in commercial.

Quantify Research Health Economics Real World Evidence Market Access The understanding of real world evidence has gained traction in recent years, with an increasing number of possible uses, from healthcare product development and approval to the final clinical decision and guidance of public policies. Adoption of rwd e in research and development (r&d) continues to accelerate, and there has been an expansion of rwd e use in commercial. With access to some of the highest quality health data worldwide, quantify research has built a reputation for delivering regulatory grade evidence grounded in nordic real world evidence. We provide a brief overview on the type and sources of real world data and the common models and approaches to utilize and analyze real world data. In this perspective, we present exemplars across various health areas that have been impacted by real world data and rwe, and we provide insights into further opportunities afforded by rwe. Explore real world evidence (rwe) and observational studies in clinical research, including study types, data sources, regulatory expectations, and best practices for generating credible rwe.
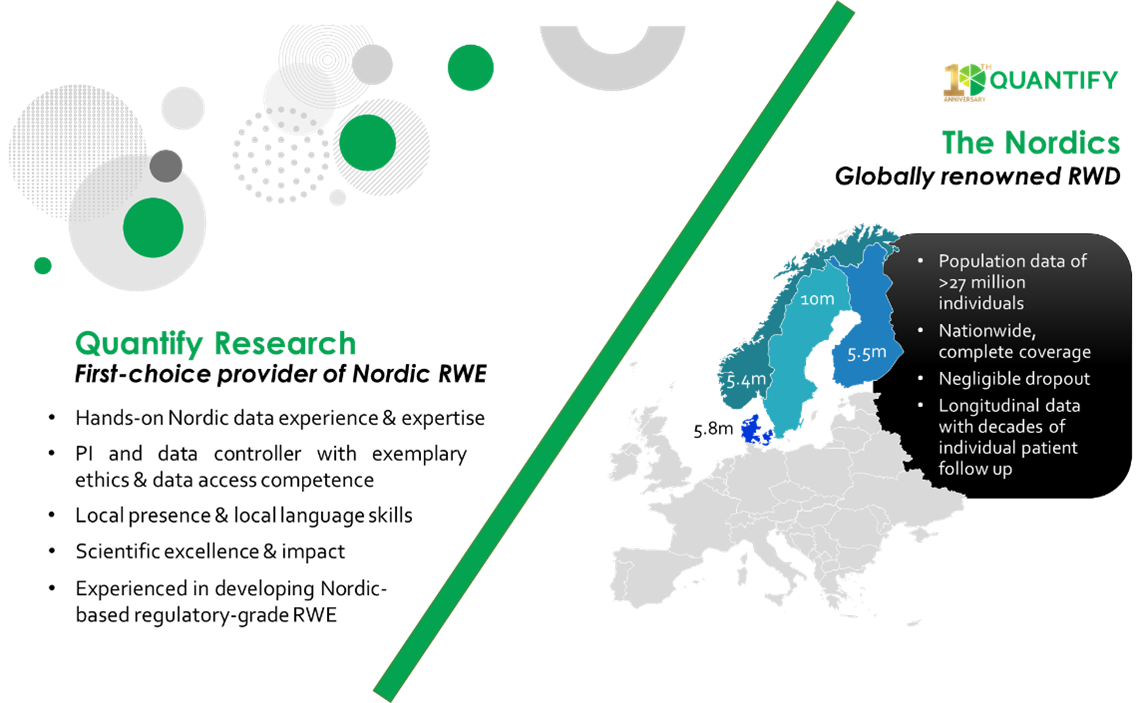
The Increasing Importance Of Real World Data Quantify Research With access to some of the highest quality health data worldwide, quantify research has built a reputation for delivering regulatory grade evidence grounded in nordic real world evidence. We provide a brief overview on the type and sources of real world data and the common models and approaches to utilize and analyze real world data. In this perspective, we present exemplars across various health areas that have been impacted by real world data and rwe, and we provide insights into further opportunities afforded by rwe. Explore real world evidence (rwe) and observational studies in clinical research, including study types, data sources, regulatory expectations, and best practices for generating credible rwe.
Comments are closed.