Real Time Release Testing Pptx

Presentation Testing 1455044600 187410 Pptx The document discusses real time release testing (rtrt) in pharmaceuticals, which evaluates product quality using process data instead of traditional end product testing. it outlines the advantages and challenges of implementing rtrt, as well as its relationship with quality by design (qbd) and process analytical technology (pat). the presentation emphasizes the paradigm shift towards. Compliance with release specifications can be demonstrated by performing a complete set of tests on the api and finished product according to the approved specifications. sometimes, alternative strategy to systematic final product testing is utilized .
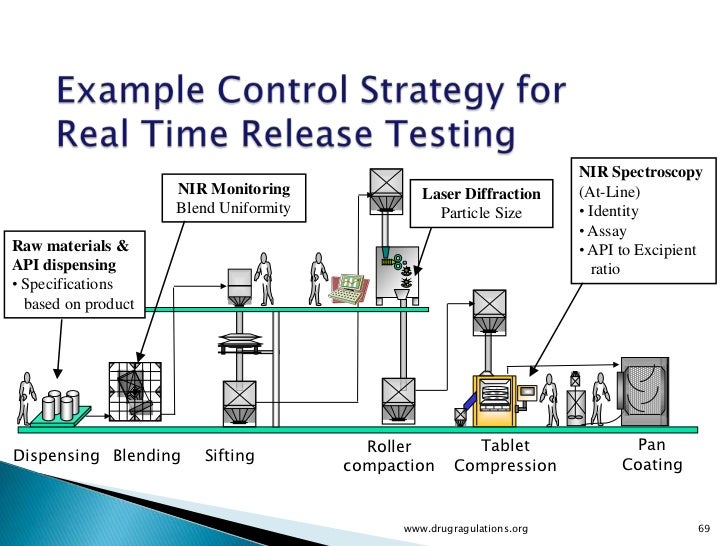
Real Time Release Testing Explore our well curated release testing presentation templates and google slides. Real time release testing (rtrt): the ability to evaluate and ensure the quality of in process and or final product based on process data, which typically include a valid combination of measured material attributes and process controls (ich q8 (r)). You can view or download release testing presentations for your school assignment or business presentation. browse for the presentations on every topic that you want. What is real time release testing? real time release testing (rtrt) is an advanced approach to biopharmaceutical manufacturing quality control that improves upon current systems.

Real Time Release Testing Pptx You can view or download release testing presentations for your school assignment or business presentation. browse for the presentations on every topic that you want. What is real time release testing? real time release testing (rtrt) is an advanced approach to biopharmaceutical manufacturing quality control that improves upon current systems. If all cqas (relevant for real time release testing) are assured by in process monitoring of parameters and or testing of materials, then end product testing might not be needed for batch release. This document outlines the requirements for applications that propose real time release testing for active substances, intermediates and finished products. it addresses the need for interaction between quality assessors and good manufacturing practice inspectors in the approval process. New ema guidance on real time release testing. this presentation provides information about real time release testing (rtrt) from the online resource drug regulations. Real time release testing (rtrt) is defined as "the ability to evaluate and ensure the quality of in process and or final drug product based on process data, which typically includes a.

Real Time Release Testing Rtrt Pat Cpv And Regulatory Expectations If all cqas (relevant for real time release testing) are assured by in process monitoring of parameters and or testing of materials, then end product testing might not be needed for batch release. This document outlines the requirements for applications that propose real time release testing for active substances, intermediates and finished products. it addresses the need for interaction between quality assessors and good manufacturing practice inspectors in the approval process. New ema guidance on real time release testing. this presentation provides information about real time release testing (rtrt) from the online resource drug regulations. Real time release testing (rtrt) is defined as "the ability to evaluate and ensure the quality of in process and or final drug product based on process data, which typically includes a.

Real Time Release Testing Pptx New ema guidance on real time release testing. this presentation provides information about real time release testing (rtrt) from the online resource drug regulations. Real time release testing (rtrt) is defined as "the ability to evaluate and ensure the quality of in process and or final drug product based on process data, which typically includes a.
Comments are closed.