Real Time Release Testing
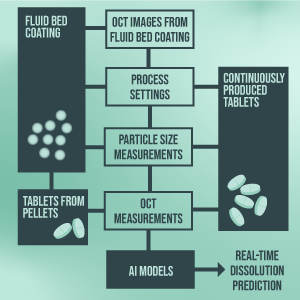
Real Time Release Testing Eccpm This document outlines the requirements for applications that propose real time release testing for active substances, intermediates and finished products. it addresses the need for interaction between quality assessors and good manufacturing practice inspectors in the approval process. This will enable real time release testing (rtrt), which redefines conventional approaches by relying solely on the process data reported by equipment or collected from sensors to predict the product quality.

Opportunities And Challenges Process Raman For The Real Time Release Real time release testing is a quality release process that facilitates the near instantaneous release of pharmaceutical drug products in batch and continuous manufacturing. Real time release testing (rtrt) is a modern quality assurance approach in pharmaceutical manufacturing that enables the release of a drug product based on real time process data rather than traditional end product testing. Regulatory submissions involving the use of continuous manufacturing (cm) 1 and or real time release testing for dissolution (rtrt d) to the united states food and drug administration (fda) were identified spanning several years. Ensure product quality while minimizing the need to take samples, review paper records, or catch deviations by implementing real time release technologies. with optimized sensing, in line monitoring, process control, and electronic workflow records, you can easily generate accurate and cohesive data during manufacturing.

Parametric Release And Real Time Release Testing Regulatory submissions involving the use of continuous manufacturing (cm) 1 and or real time release testing for dissolution (rtrt d) to the united states food and drug administration (fda) were identified spanning several years. Ensure product quality while minimizing the need to take samples, review paper records, or catch deviations by implementing real time release technologies. with optimized sensing, in line monitoring, process control, and electronic workflow records, you can easily generate accurate and cohesive data during manufacturing. What is real time release testing? real time release testing (rtrt) is an advanced approach to biopharmaceutical manufacturing quality control that improves upon current systems. Real‑time release testing (rtrt) is an approach where a batch is released based on in‑process and or real‑time data demonstrating that it meets predefined quality criteria, rather than relying primarily on end‑product laboratory tests. Learn how sensors and control strategies enable real time release testing (rtrt) for biologic drugs. rtrt is the ability to evaluate and ensure product quality based on process data, which can improve safety, efficacy, and productivity. Real time release testing (rtrt): the ability to evaluate and ensure the quality of in process and or final product based on process data, which typically include a valid combination of measured material attributes and process controls (ich q8 (r)).

Real Time Release Testing Scientific Guideline European Medicines What is real time release testing? real time release testing (rtrt) is an advanced approach to biopharmaceutical manufacturing quality control that improves upon current systems. Real‑time release testing (rtrt) is an approach where a batch is released based on in‑process and or real‑time data demonstrating that it meets predefined quality criteria, rather than relying primarily on end‑product laboratory tests. Learn how sensors and control strategies enable real time release testing (rtrt) for biologic drugs. rtrt is the ability to evaluate and ensure product quality based on process data, which can improve safety, efficacy, and productivity. Real time release testing (rtrt): the ability to evaluate and ensure the quality of in process and or final product based on process data, which typically include a valid combination of measured material attributes and process controls (ich q8 (r)).
Comments are closed.