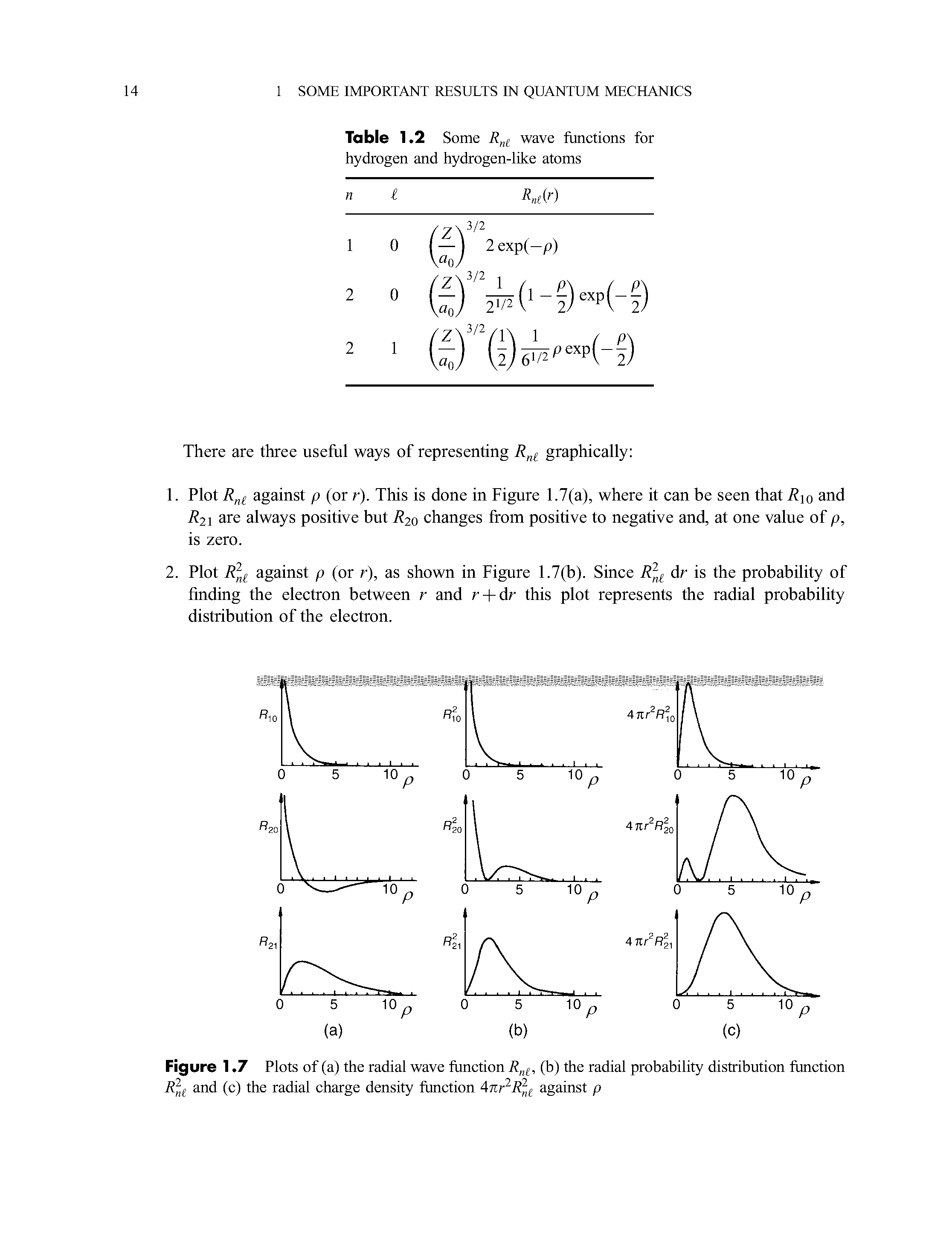
Radial Probability Distribution Functions

Radial Probability Distribution Function With Anno Pdf In statistical mechanics, the radial distribution function, (or pair correlation function) in a system of particles (atoms, molecules, colloids, etc.), describes how density varies as a function of distance from a reference particle. The radial distribution function (rdf), denoted as g (r), is a fundamental concept in statistical mechanics, that describes the probability of finding a particle at a distance r from a reference particle in a homogeneous and isotropic system [1].

Radial Probability Distribution Function Big Chemical Encyclopedia The radial distribution function (rdf) defines the probability of finding a particle at distance r from another tagged particle. here, the distance r is between the oxygen atoms of two water molecules. The radial portion of the wavefunction really only tells us if there is high or low probability at various distances from the nucleus (possible radii for the electrons). It is also known as radial probability density function, it is given by 4πr 2 r 2nl (r). in the graphs shown in question, ψ 2 is shown instead of r 2nl (r). it gives idea about the distribution of electron density at a radial distance around the nucleus without considering the direction or angle. After multiplying the “probability distribution function” with “volume element”, the expression for the “radial distribution function” can be formulated. mathematically, we can say that. it is obvious from the equation (442) that probability will become zero if we put r = 0 (4 2 =0).
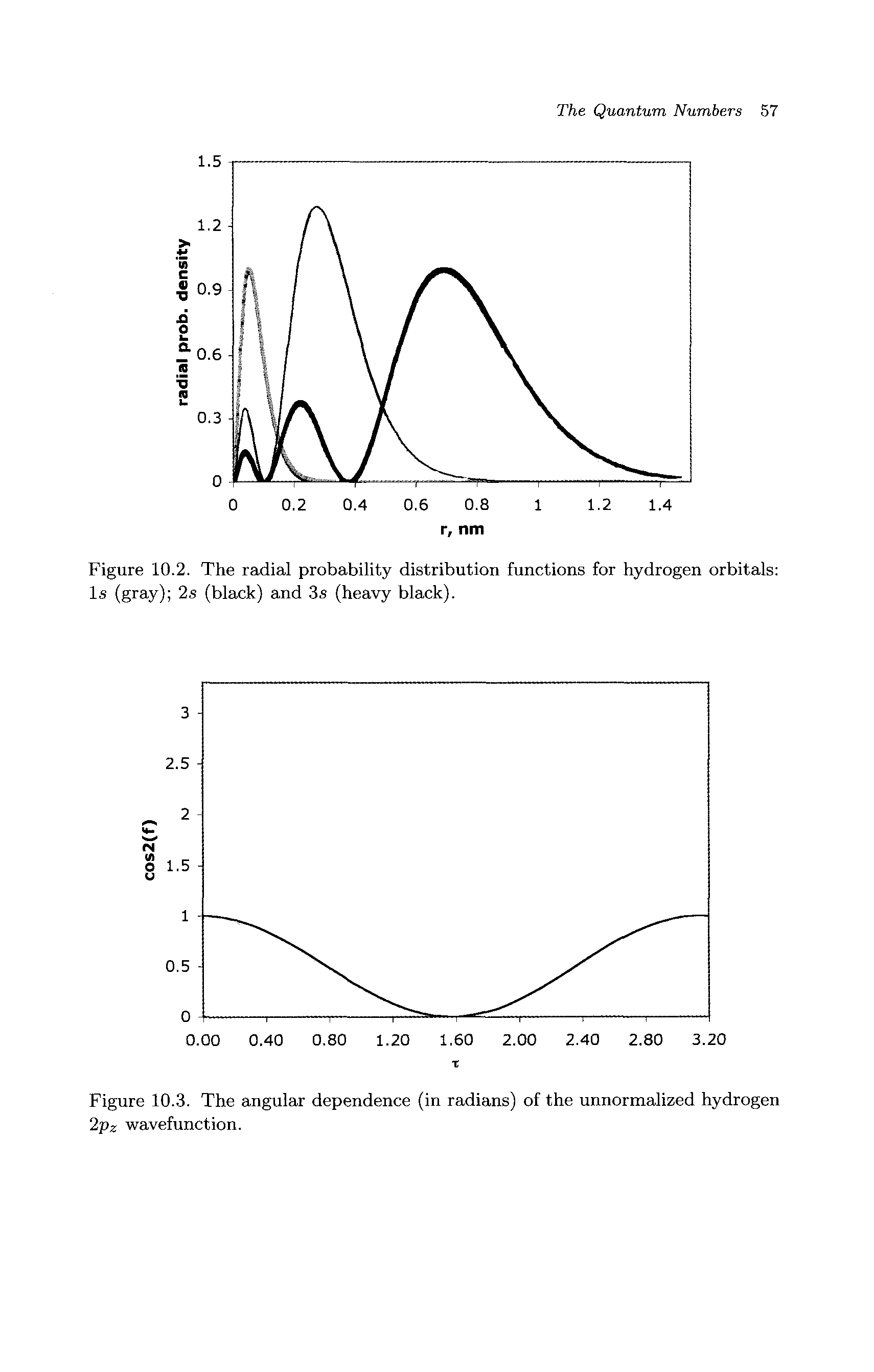
Radial Probability Distribution Function Big Chemical Encyclopedia It is also known as radial probability density function, it is given by 4πr 2 r 2nl (r). in the graphs shown in question, ψ 2 is shown instead of r 2nl (r). it gives idea about the distribution of electron density at a radial distance around the nucleus without considering the direction or angle. After multiplying the “probability distribution function” with “volume element”, the expression for the “radial distribution function” can be formulated. mathematically, we can say that. it is obvious from the equation (442) that probability will become zero if we put r = 0 (4 2 =0). The radial distribution function is most commonly used in gasses, liquids, and solutions, since it can be used to calculate thermodynamic properties such as the internal energy and pressure of the system. A comprehensive guide to radial distribution function (rdf) analysis in statistical mechanics and molecular simulation, covering its principles, applications, and best practices. If we want to find the probability of finding at a given r independent of angular distribution, it is convenient to define a quantity called the radial distribution function p (r) which is defined as. These functions give the density probability for an atom of the α species to have a neighbor of the β species at a given distance r. the example features ges 2 glass.
Radial Probability Distribution Function Big Chemical Encyclopedia The radial distribution function is most commonly used in gasses, liquids, and solutions, since it can be used to calculate thermodynamic properties such as the internal energy and pressure of the system. A comprehensive guide to radial distribution function (rdf) analysis in statistical mechanics and molecular simulation, covering its principles, applications, and best practices. If we want to find the probability of finding at a given r independent of angular distribution, it is convenient to define a quantity called the radial distribution function p (r) which is defined as. These functions give the density probability for an atom of the α species to have a neighbor of the β species at a given distance r. the example features ges 2 glass.

Radial Probability Distribution Function Big Chemical Encyclopedia If we want to find the probability of finding at a given r independent of angular distribution, it is convenient to define a quantity called the radial distribution function p (r) which is defined as. These functions give the density probability for an atom of the α species to have a neighbor of the β species at a given distance r. the example features ges 2 glass.
Comments are closed.