Radial Probability Density Radial Distribution Function An Overview

Radial Probability Distribution Function With Anno Pdf In statistical mechanics, the radial distribution function, (or pair correlation function) in a system of particles (atoms, molecules, colloids, etc.), describes how density varies as a function of distance from a reference particle. Rdf provides insights into the structure of liquids and disordered materials. the relationship between rdfs and thermodynamic properties is explored. rdfs play a crucial role in understanding intermolecular forces.

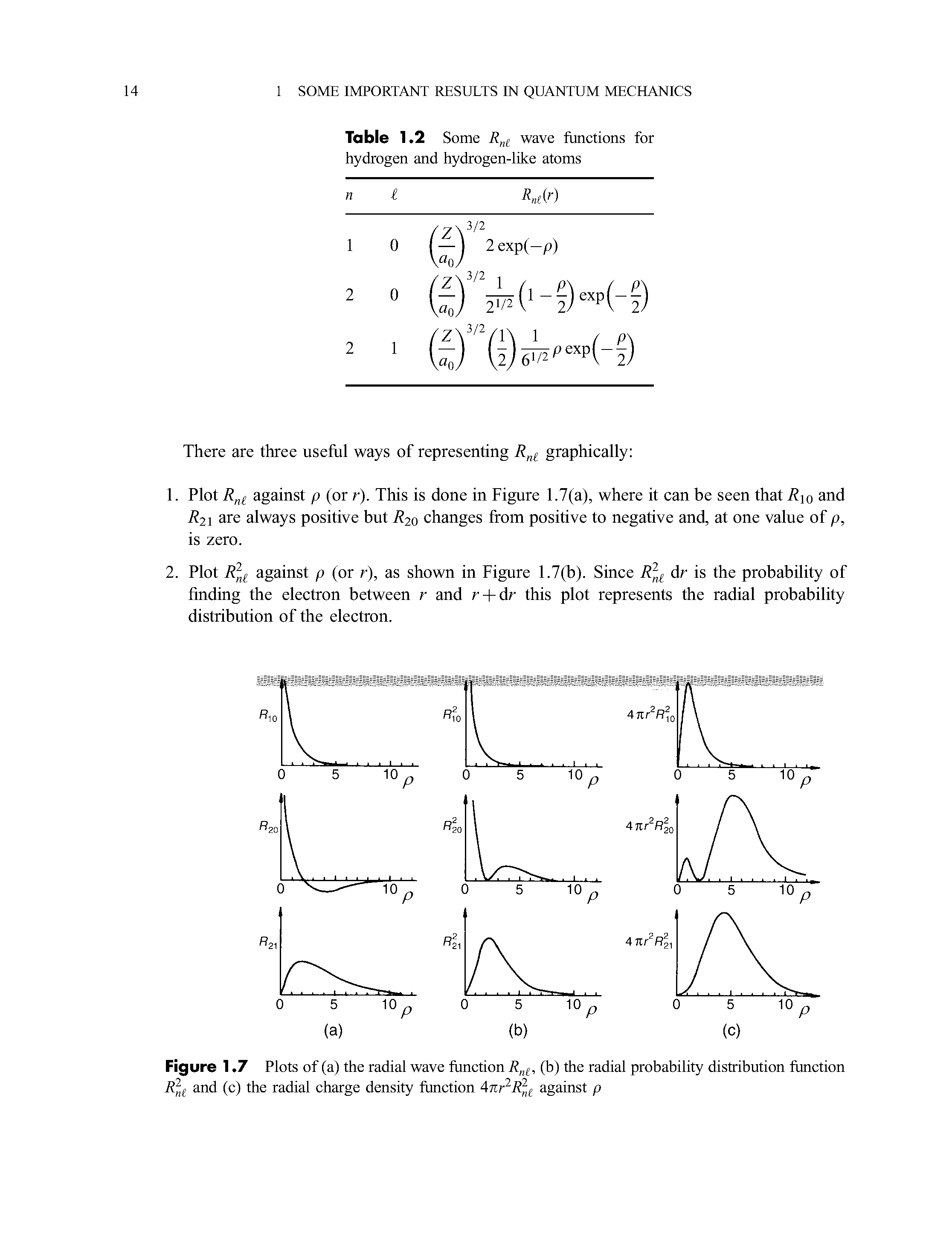
Radial Probability Distribution Function Big Chemical Encyclopedia It is also known as radial probability density function, it is given by 4πr 2 r 2nl (r). in the graphs shown in question, ψ 2 is shown instead of r 2nl (r). it gives idea about the distribution of electron density at a radial distance around the nucleus without considering the direction or angle. In the radial distribution plots, we assume that the probability of finding the particle at a distance r from the nucleus depends not only upon the density of electron wave but also varies with the magnitude of the volume of the spherical shell of dr thickness at the same distance. A comprehensive guide to radial distribution function (rdf) analysis in statistical mechanics and molecular simulation, covering its principles, applications, and best practices. Formal approach: radial distribution function (rdf) the radial distribution function is defined as overall density of atoms (volume) g ( r ) = ρ ( r ) ρ local density provides information about the density of atoms at a given radius r; ρ(r) is the local density of atoms.
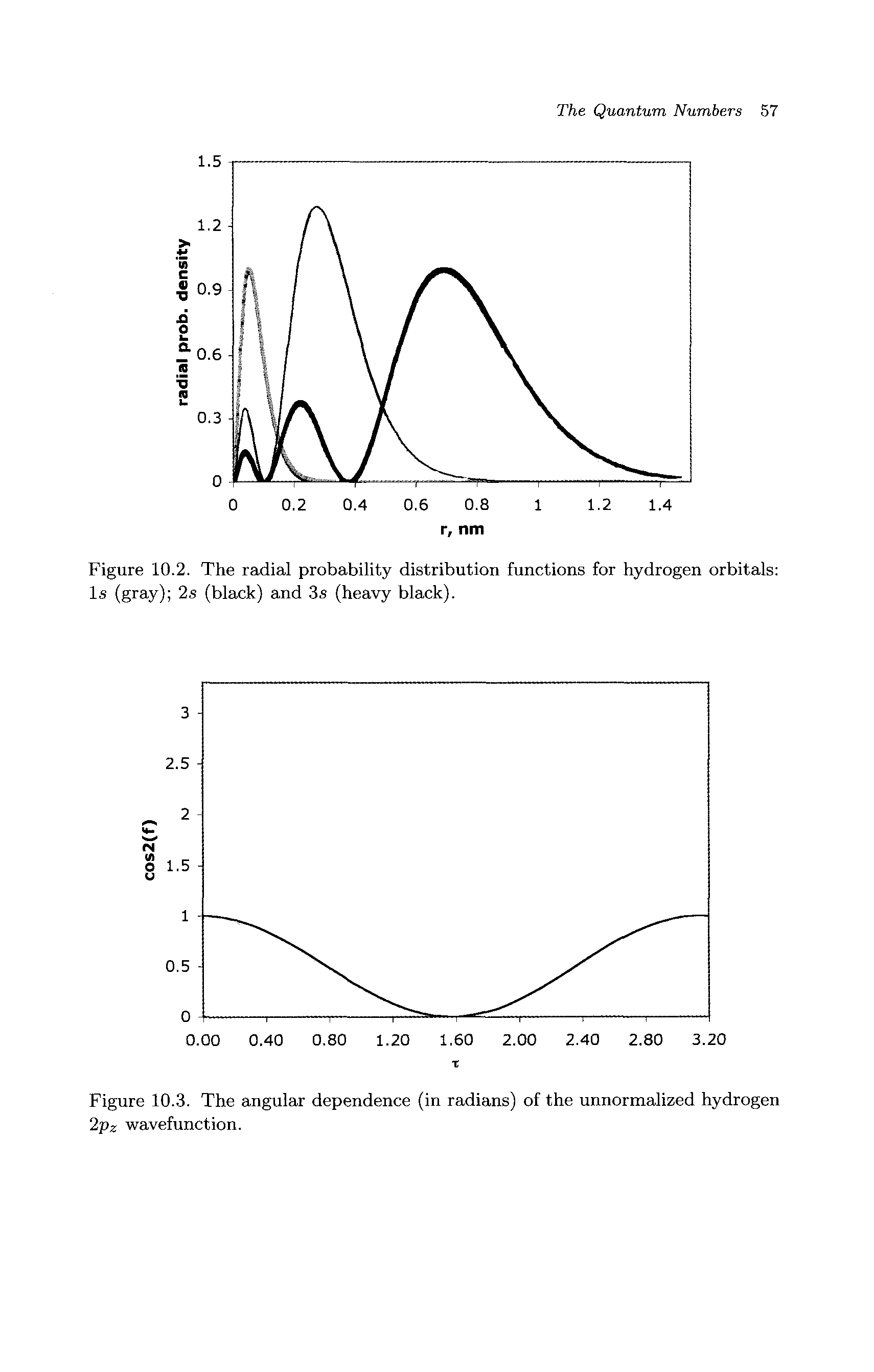
Radial Probability Distribution Function Big Chemical Encyclopedia A comprehensive guide to radial distribution function (rdf) analysis in statistical mechanics and molecular simulation, covering its principles, applications, and best practices. Formal approach: radial distribution function (rdf) the radial distribution function is defined as overall density of atoms (volume) g ( r ) = ρ ( r ) ρ local density provides information about the density of atoms at a given radius r; ρ(r) is the local density of atoms. The radial probability density is the probability of finding an electron at a distance r from the nucleus. it is calculated by adding together the probabilities of an electron being at all points on a series of spherical shells of radius r1, r2, r3,…, rx − 1, rx. Where r(r) is the radial part of the probability distribution function. the radial distribution gives the probability density at a distance r from the nucleus. for example, we can use the 1s orbital and find out the distance rmax from the nucleus where the electron is most likely to be found by p (r) ∝ r2e−2r a0 using the condition for. Compute the radial distribution function using the formula given above. let’s consider an example to illustrate how to compute the radial distribution function for a simple system of particles. At very long range the radial distribution function tends to 1. the radial distribution function can be thought of as the ratio of local density to global density.
Radial Probability Distribution Function Big Chemical Encyclopedia The radial probability density is the probability of finding an electron at a distance r from the nucleus. it is calculated by adding together the probabilities of an electron being at all points on a series of spherical shells of radius r1, r2, r3,…, rx − 1, rx. Where r(r) is the radial part of the probability distribution function. the radial distribution gives the probability density at a distance r from the nucleus. for example, we can use the 1s orbital and find out the distance rmax from the nucleus where the electron is most likely to be found by p (r) ∝ r2e−2r a0 using the condition for. Compute the radial distribution function using the formula given above. let’s consider an example to illustrate how to compute the radial distribution function for a simple system of particles. At very long range the radial distribution function tends to 1. the radial distribution function can be thought of as the ratio of local density to global density.

Radial Probability Distribution Function Big Chemical Encyclopedia Compute the radial distribution function using the formula given above. let’s consider an example to illustrate how to compute the radial distribution function for a simple system of particles. At very long range the radial distribution function tends to 1. the radial distribution function can be thought of as the ratio of local density to global density.
Comments are closed.