Quantum Chemistry 2 1 Classical Wave Equation
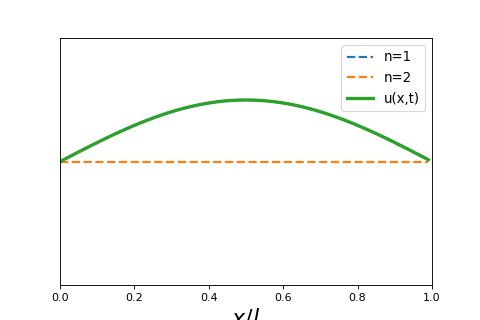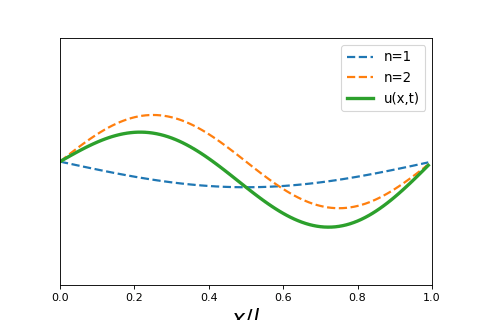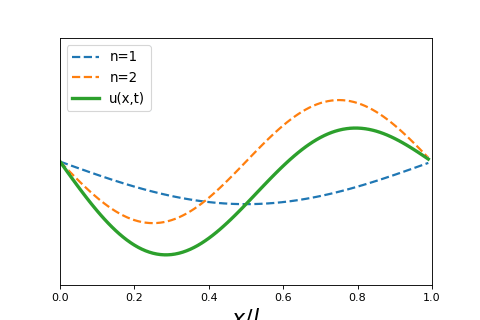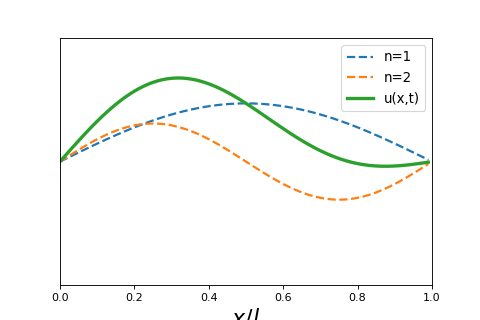
4 5 2 Wave Particle Duality And The Classical Wave Equation Short lecture on the classical wave equation. the classical wave equation relates the second derivative in space to the second derivative in time of a wave function. Wave equation overview: the wave equation, a partial differential equation (pde), fully describes the spatial and temporal evolution of waves. linearity of the wave equation: the wave equation is linear, meaning that any linear combination of solutions is also a solution.
4 5 2 Wave Particle Duality And The Classical Wave Equation In this lecture we will review and solve the classical wave equation, a partial differential equation that governs the spatial and temporal behavior of waves. Equation 8.1.2 is called the classical wave equation in one dimension and is a linear partial differential equation. it tells us how the displacement u can change as a function of position and time and the function. Video answers for all textbook questions of chapter 2, the classical wave equation, quantum chemistry by numerade. The schrödinger equation gives the evolution over time of the wave function, the quantum mechanical characterization of an isolated physical system. the equation was postulated by schrödinger based on a postulate of louis de broglie that all matter has an associated matter wave.
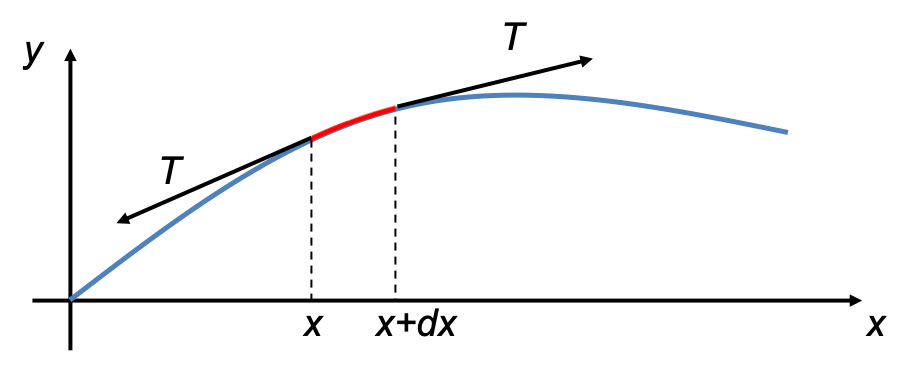
1 D Classical Wave Equation Mono Mole Video answers for all textbook questions of chapter 2, the classical wave equation, quantum chemistry by numerade. The schrödinger equation gives the evolution over time of the wave function, the quantum mechanical characterization of an isolated physical system. the equation was postulated by schrödinger based on a postulate of louis de broglie that all matter has an associated matter wave. Exercises set 02. classical wave equation free download as pdf file (.pdf), text file (.txt) or read online for free. exercises guide of quantum chemistry. Let’s begin by reminding ourselves of the wave equation for waves on a taut string, stretched between x = 0 and x = l, tension t newtons, density ρ kg meter. In quantum mechanics you will see wavepackets that exhibit motion, breathing, dephasing, and rephasing. the “center of the wavepacket” will follow a trajectory that obeys newton’s laws of motion. After the failure of the bohr atomic model to comply with the heisenberg’s uncertainty principle and dual character proposed by louis de broglie in 1924, an austrian physicist erwin schrodinger developed his legendary equation by making the use of wave particle duality and classical wave equation.

Ppt Classical Wave Equation Exercises set 02. classical wave equation free download as pdf file (.pdf), text file (.txt) or read online for free. exercises guide of quantum chemistry. Let’s begin by reminding ourselves of the wave equation for waves on a taut string, stretched between x = 0 and x = l, tension t newtons, density ρ kg meter. In quantum mechanics you will see wavepackets that exhibit motion, breathing, dephasing, and rephasing. the “center of the wavepacket” will follow a trajectory that obeys newton’s laws of motion. After the failure of the bohr atomic model to comply with the heisenberg’s uncertainty principle and dual character proposed by louis de broglie in 1924, an austrian physicist erwin schrodinger developed his legendary equation by making the use of wave particle duality and classical wave equation.
Comments are closed.