Quality Control Processes For Precise And Accurate Microbial Next

Quality Control Processes For Precise And Accurate Microbial Next The ngs diagnostic process is subjected to rigorous multistep quality controls to guarantee that external contaminants are excluded, closely related species are differentiated, and organisms present in the clinical sample are accurately identified. This article explores the importance of quality control in microbiological testing, the key practices for ensuring consistency and accuracy, and the regulatory frameworks that labs must adhere to.

Ensuring Excellence Through Standardization A Comprehensive Look At A laboratory quality management system is a systematic, integrated set of activities to establish and control the work processes from preanalytical through postanalytical processes, manage resources, conduct evaluations, and make continual improvements to ensure consistent quality results. Discover the latest techniques, tools, and best practices for quality assurance in laboratory microbiology to enhance the accuracy and reliability of test results. Meticulous attention to laboratory layout, staff training, documentation, quality control, and safety procedures helps uphold high accuracy, efficiency, and compliance standards. regular audits and adherence to regulatory guidelines also play essential roles in maintaining these standards. Process control is an essential element of the quality management system, and refers to control of the activities employed in the handling of samples and examination processes in order to ensure accurate and reliable testing.

Critical Terminology For Microbial Control Meticulous attention to laboratory layout, staff training, documentation, quality control, and safety procedures helps uphold high accuracy, efficiency, and compliance standards. regular audits and adherence to regulatory guidelines also play essential roles in maintaining these standards. Process control is an essential element of the quality management system, and refers to control of the activities employed in the handling of samples and examination processes in order to ensure accurate and reliable testing. The quality control (qc) practice in a microbiology laboratory is a bit different than what we have covered till now. this chapter provides an overview of quality control specific to microbiology. Quality control focuses on operational activities that detect and correct defects in products or analytical results. it emphasizes product testing, inspection, measurement, and validation against established specifications. To ensure effective quality control in microbiological testing and avoid common mistakes, you should provide detailed sops (standard operating procedures) with instructions on how to correctly carry out tests, safely manage samples and accurately analyse results. The process control quality system essential (qse) details the management of processes throughout all phases of testing (preexamination, examination, and post examination).
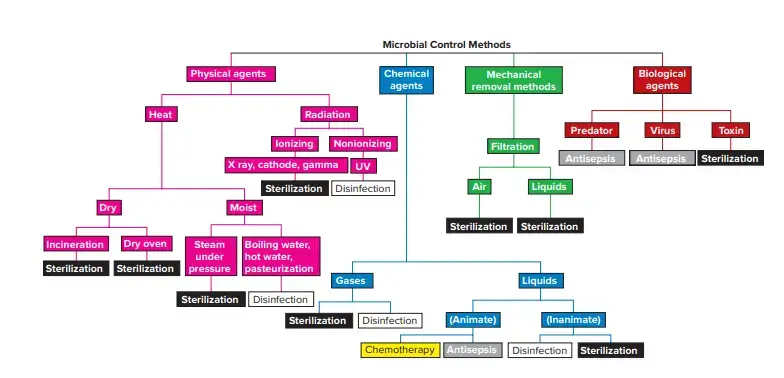
Microbial Control Methods Biology Notes Online The quality control (qc) practice in a microbiology laboratory is a bit different than what we have covered till now. this chapter provides an overview of quality control specific to microbiology. Quality control focuses on operational activities that detect and correct defects in products or analytical results. it emphasizes product testing, inspection, measurement, and validation against established specifications. To ensure effective quality control in microbiological testing and avoid common mistakes, you should provide detailed sops (standard operating procedures) with instructions on how to correctly carry out tests, safely manage samples and accurately analyse results. The process control quality system essential (qse) details the management of processes throughout all phases of testing (preexamination, examination, and post examination).

Microbial Quality Control In Manufacturing Of Medicinal Products To ensure effective quality control in microbiological testing and avoid common mistakes, you should provide detailed sops (standard operating procedures) with instructions on how to correctly carry out tests, safely manage samples and accurately analyse results. The process control quality system essential (qse) details the management of processes throughout all phases of testing (preexamination, examination, and post examination).

Improved Production And Quality Control Processes
Comments are closed.