Process Verification And Validation Kuglqu
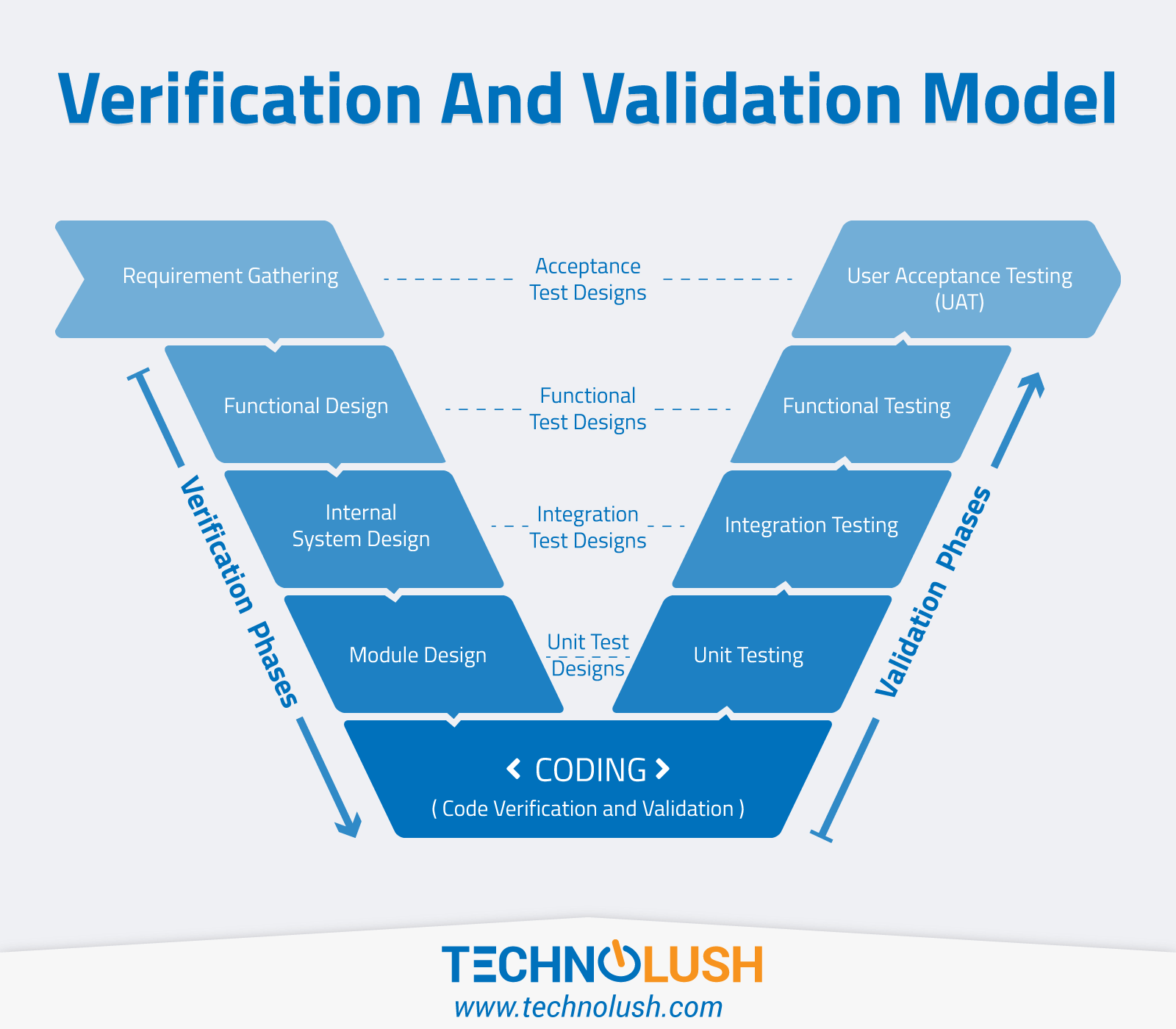
Validation And Verification Process Qgwr In the following sections, we describe general considerations for process validation, the recommended stages of process validation, and specific activities for each stage in the product lifecycle. A look at what process verification and process validation refer to, and when you should use each of them in medical device manufacturing.

Process Verification And Validation Kuglqu The guideline is brought into line with ich q8, q9 and q10 documents and the possibility to use continuous process verification in addition to, or instead of, traditional process validation described in the previous guideline has been added and is encouraged. Understand the critical differences between process validation and verification in quality management systems (qms). learn about their benefits, challenges, types, stages, and the role of cmms. Whereas process verification determines whether a process can produce a single product that meets specifications and requirements, process validation testing is concerned with ensuring that the process can accomplish this more than once. Validation provides confidence that a process will perform consistently, while verification confirms that specific instances did perform as expected. both are necessary for a robust quality assurance program.
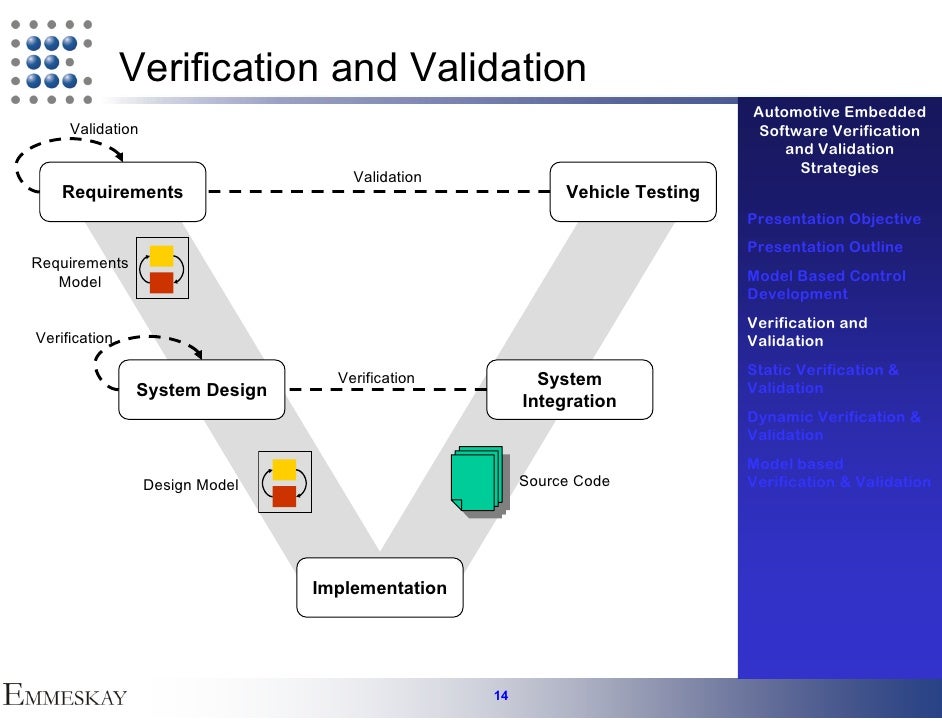
Process Verification And Validation Kuglqu Whereas process verification determines whether a process can produce a single product that meets specifications and requirements, process validation testing is concerned with ensuring that the process can accomplish this more than once. Validation provides confidence that a process will perform consistently, while verification confirms that specific instances did perform as expected. both are necessary for a robust quality assurance program. Process verification and validation are crucial components of quality manufacturing. here we discuss their similarities and differences. For a medical device manufacturer, one of the most challenging aspects of compliance is understanding whether a manufacturing process must be verif. Performing process verification and validation can be challenging, but experts at tsq & e are ready to help. as outlined in this guide, these processes are intricate but essential for regulatory compliance. Process validation lifecycle – learn the three stages: process design, process qualification, and continued process verification (cpv). equipment & facility qualification – understand iq, oq, pq processes and their role in ensuring equipment and facility compliance.

Process Verification Vs Validation What S The Difference Process verification and validation are crucial components of quality manufacturing. here we discuss their similarities and differences. For a medical device manufacturer, one of the most challenging aspects of compliance is understanding whether a manufacturing process must be verif. Performing process verification and validation can be challenging, but experts at tsq & e are ready to help. as outlined in this guide, these processes are intricate but essential for regulatory compliance. Process validation lifecycle – learn the three stages: process design, process qualification, and continued process verification (cpv). equipment & facility qualification – understand iq, oq, pq processes and their role in ensuring equipment and facility compliance.

Process Verification Vs Process Validation What S The Difference Performing process verification and validation can be challenging, but experts at tsq & e are ready to help. as outlined in this guide, these processes are intricate but essential for regulatory compliance. Process validation lifecycle – learn the three stages: process design, process qualification, and continued process verification (cpv). equipment & facility qualification – understand iq, oq, pq processes and their role in ensuring equipment and facility compliance.

Process Validation For Medical Devices Process Validation 40 Off
Comments are closed.