Process Validation Process Steps Quvly

Process Validation Process Steps Quvly In the following sections, we describe general considerations for process validation, the recommended stages of process validation, and specific activities for each stage in the product lifecycle. This guideline presents elements for consideration during the validation of analytical procedures included as part of registration applications. analytical procedure validation forms a part of the analytical procedure lifecycle, as described within ich q14 analytical procedure development. ich q2(r2) provides guidance on selection and evaluation of the various validation tests for analytical.

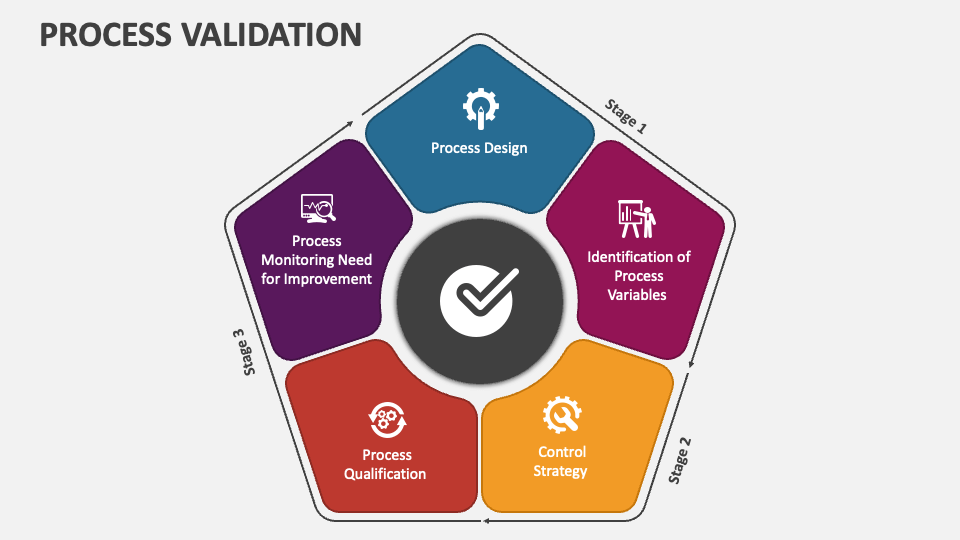
Process Validation Procedure 4easyreg This article delves into the process validation lifecycle model, examining the three pivotal stages: stage 1 – process design, stage 2 – process performance qualification (ppq), and stage 3 – ongoing process verification. This guidance provides a comprehensive framework and recommendations for the validation of pharmaceutical processes, including the principles of process validation, stages of process validation, and statistical tools for data analysis. The process validation lifecycle is generally broken down into three distinct phases: process design, process qualification and continued process verification. each phase serves a unique purpose in ensuring the final product consistently meets quality standards. It consists of three stages — process design, process performance qualification (ppq), and continued process verification (cpv). this evolution underscores fda’s commitment to science and risk based decision making.

Applying A Validation Process In 5 Steps Qseqnn The process validation lifecycle is generally broken down into three distinct phases: process design, process qualification and continued process verification. each phase serves a unique purpose in ensuring the final product consistently meets quality standards. It consists of three stages — process design, process performance qualification (ppq), and continued process verification (cpv). this evolution underscores fda’s commitment to science and risk based decision making. Nce qualification · continued process verification as defined by the us fda process validation guidance [1], process validation is the collection and evaluation of data, from the process design stage through com mercial production, which establishes scientific evidence that a process is. This comprehensive guide provides you with a complete checklist and step by step approach to validate any process effectively. you’ll learn how to ensure consistent quality and regulatory compliance from day one, using methods that have helped thousands of companies avoid expensive mistakes. This guideline presents a discussion of elements for consideration during the validation of analytical procedures included as part of registration applications submitted within the ich member regulatory authorities. it provides guidance and recommendations on how to derive and evaluate the various validation tests for each analytical procedure and serves as a collection of terms, and their. Process validation lifecycle – learn the three stages: process design, process qualification, and continued process verification (cpv). equipment & facility qualification – understand iq, oq, pq processes and their role in ensuring equipment and facility compliance.
Process Validation Powerpoint Presentation Slides Ppt Template Nce qualification · continued process verification as defined by the us fda process validation guidance [1], process validation is the collection and evaluation of data, from the process design stage through com mercial production, which establishes scientific evidence that a process is. This comprehensive guide provides you with a complete checklist and step by step approach to validate any process effectively. you’ll learn how to ensure consistent quality and regulatory compliance from day one, using methods that have helped thousands of companies avoid expensive mistakes. This guideline presents a discussion of elements for consideration during the validation of analytical procedures included as part of registration applications submitted within the ich member regulatory authorities. it provides guidance and recommendations on how to derive and evaluate the various validation tests for each analytical procedure and serves as a collection of terms, and their. Process validation lifecycle – learn the three stages: process design, process qualification, and continued process verification (cpv). equipment & facility qualification – understand iq, oq, pq processes and their role in ensuring equipment and facility compliance.
Comments are closed.