Process Deviations Quality Collaboration By Design
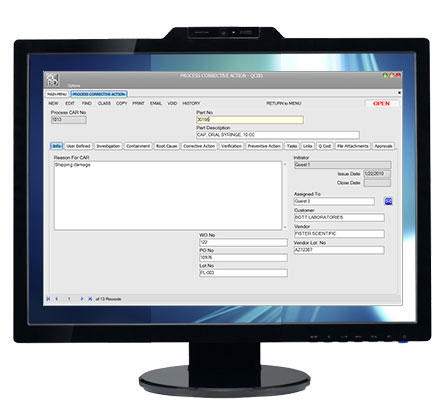
Process Deviations Quality Collaboration By Design The qcbd process deviation module manages and documents departure from standard practices and standard procedures typically used in fda and iso documentation. regardless of how well written your procedures and work instructions are, it is often necessary to deviate from those documented practices. Quality by design (qbd) is a systematic approach to development that starts with research derived objectives and promotes product and process knowledge, based on sound science and quality risk management.

11 Process Deviations Icon Images Stock Photos Vectors Shutterstock Analytical quality through design is grounded in the core qbd principles, which focus on clearly establishing how product attributes relate to the process parameters that influence them. Such “process deviations” from approved specifications can range from minor to major and potentially compromise standards if not promptly addressed. identifying, investigating, and resolving deviations is thus critical for quality management. Deviation denotes the fact that a process execution is not conform to the nor mative process model. Learn how quality by design (qbd) ensures consistent pharmaceutical quality through process understanding, risk management, and regulatory alignment.

Deviations And Process Improvements Learngxp Accredited Online Life Deviation denotes the fact that a process execution is not conform to the nor mative process model. Learn how quality by design (qbd) ensures consistent pharmaceutical quality through process understanding, risk management, and regulatory alignment. Introduction quality by design (qbd) is fundamentally transforming bioprocess development by offering a departure from traditional empirical methodologies towards a systematic, science and risk based strategy. this sophisticated approach mandates an early and deep understanding of critical process parameters (cpps) and critical quality attributes (cqas), enabling the establishment of a well. Process deviations represent formal gmp events that signal potential loss of control, not routine execution errors. a process deviation refers to any unplanned departure from an approved manufacturing or quality procedure that may affect product quality, patient safety, or regulatory compliance. A quality by design study for an immediate release tablet platform: examining the relative impact of active pharmaceutical ingredient properties, processing methods, and excipient variability on drug product quality attributes. Quality by design (qbd) is a systematic and science based approach that emphasizes the proactive design and development of pharmaceutical products and processes. this abstract provides an.
Collaboration Design Introduction quality by design (qbd) is fundamentally transforming bioprocess development by offering a departure from traditional empirical methodologies towards a systematic, science and risk based strategy. this sophisticated approach mandates an early and deep understanding of critical process parameters (cpps) and critical quality attributes (cqas), enabling the establishment of a well. Process deviations represent formal gmp events that signal potential loss of control, not routine execution errors. a process deviation refers to any unplanned departure from an approved manufacturing or quality procedure that may affect product quality, patient safety, or regulatory compliance. A quality by design study for an immediate release tablet platform: examining the relative impact of active pharmaceutical ingredient properties, processing methods, and excipient variability on drug product quality attributes. Quality by design (qbd) is a systematic and science based approach that emphasizes the proactive design and development of pharmaceutical products and processes. this abstract provides an.

Deviation Management Software Product Deviations A quality by design study for an immediate release tablet platform: examining the relative impact of active pharmaceutical ingredient properties, processing methods, and excipient variability on drug product quality attributes. Quality by design (qbd) is a systematic and science based approach that emphasizes the proactive design and development of pharmaceutical products and processes. this abstract provides an.

Deviation Management Software Product Deviations
Comments are closed.