Practice Problems On Balancing Equations Excess Limiting Reactants

Lesson Vi Limiting And Excess Reactants Pdf Mole Unit Chemical Problem #4: interpret reactions in terms of representative particles, then write balanced chemical equations and compare with your results. determine limiting and excess reagent and the amount of unreacted excess reactant. It includes four questions that range from basic identification of limiting reactants to more complex calculations involving mass, percent yield, and concentration. each question is based on balanced chemical equations and requires students to apply their knowledge of stoichiometry.

Balancing Equations Limiting Reactants Vocabulary Worksheet Tpt Give the balanced chemical equation for the reaction between cacl2 and hcl, states included, where products are either in aqueous, liquid or gaseous states. caco3(s) 2hcl → cacl2(aq) co2(g) h2o(l). Write the balanced equation for the reaction given below: co2 h2o. a) if 16.4 l of c2h6 reacts with 0.980 mol of o2 how many liters of carbon dioxide gas will be produced? b) how many oxygen atoms will be in this sample of carbon dioxide? c) how many moles of the excess reactant will be left over?. This is a comprehensive, end of chapter set of practice problems on stoichiometry that covers balancing chemical equations, mole ratio calculations, limiting reactants, and percent yield concepts. Practice stoichiometry with this worksheet on limiting and excess reactants. includes s'mores & race car analogies.

Balancing Equations Limiting Reactants Flipped Classroom Lesson Bundle This is a comprehensive, end of chapter set of practice problems on stoichiometry that covers balancing chemical equations, mole ratio calculations, limiting reactants, and percent yield concepts. Practice stoichiometry with this worksheet on limiting and excess reactants. includes s'mores & race car analogies. This online quiz is intended to give you extra practice in performing stoichiometric conversions, including limiting reagent and percent yield problems. this quiz aligns with ap® chemistry learning objective 4.5 stoichiometry; and ngss standard hs ps1 7. Directions: determine the limiting and excess reactants for each of the problems below. This worksheet with answers aims to provide practice problems to help students grasp the concept of limiting and excess reactants. it includes a set of chemical equations and asks students to determine the limiting reactant, calculate the amount of product formed, and identify the excess reactant. Practice problems on balancing equations, excess and limiting reactants, empirical and molecular formula.

Chemistry Limiting And Excess Reactants Practice 2 Tpt This online quiz is intended to give you extra practice in performing stoichiometric conversions, including limiting reagent and percent yield problems. this quiz aligns with ap® chemistry learning objective 4.5 stoichiometry; and ngss standard hs ps1 7. Directions: determine the limiting and excess reactants for each of the problems below. This worksheet with answers aims to provide practice problems to help students grasp the concept of limiting and excess reactants. it includes a set of chemical equations and asks students to determine the limiting reactant, calculate the amount of product formed, and identify the excess reactant. Practice problems on balancing equations, excess and limiting reactants, empirical and molecular formula.
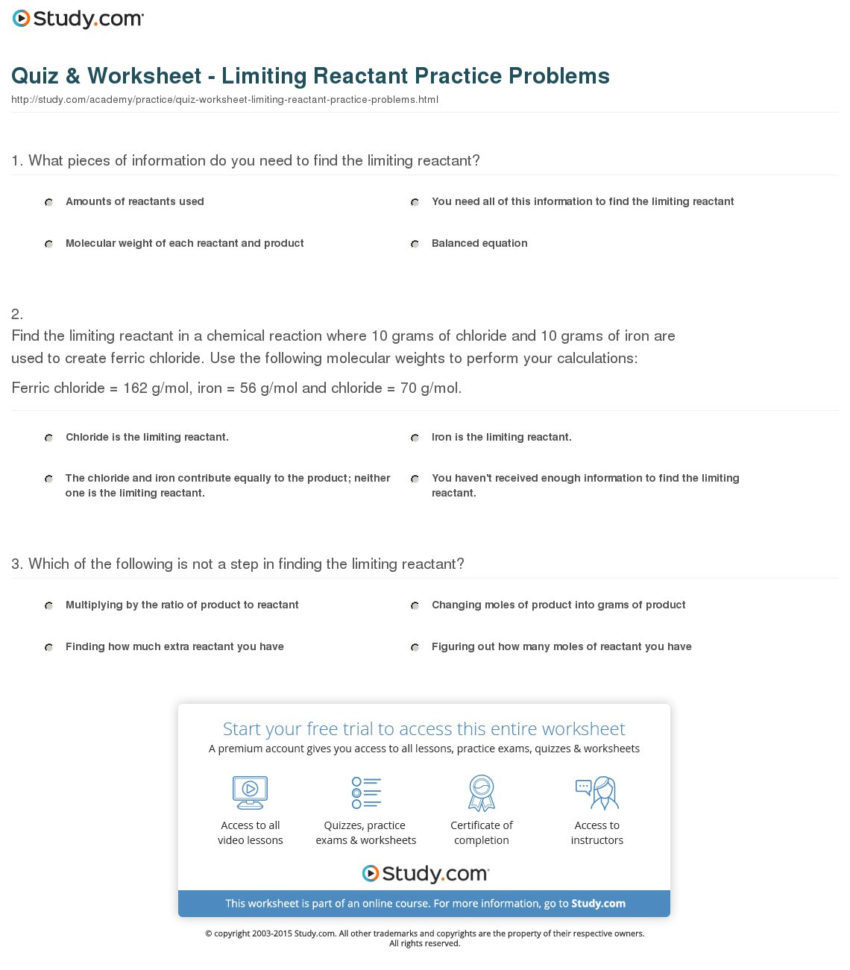
Limiting And Excess Reactants Worksheet Db Excel This worksheet with answers aims to provide practice problems to help students grasp the concept of limiting and excess reactants. it includes a set of chemical equations and asks students to determine the limiting reactant, calculate the amount of product formed, and identify the excess reactant. Practice problems on balancing equations, excess and limiting reactants, empirical and molecular formula.
Comments are closed.