Phases Of Clinical Trials

Phases Of Clinical Trials Colpitts Clinical Clinical trials are the scientific cornerstone of modern medicine, providing the evidence base for the safety and efficacy of new drugs and therapies. they are traditionally organized into four successive phases – i, ii, iii, and iv – each with distinct objectives, designs, and regulatory roles. Clinical trials refer to medical research that involves testing new treatments on people. the four phases help to ensure the trial's safety and effectiveness.
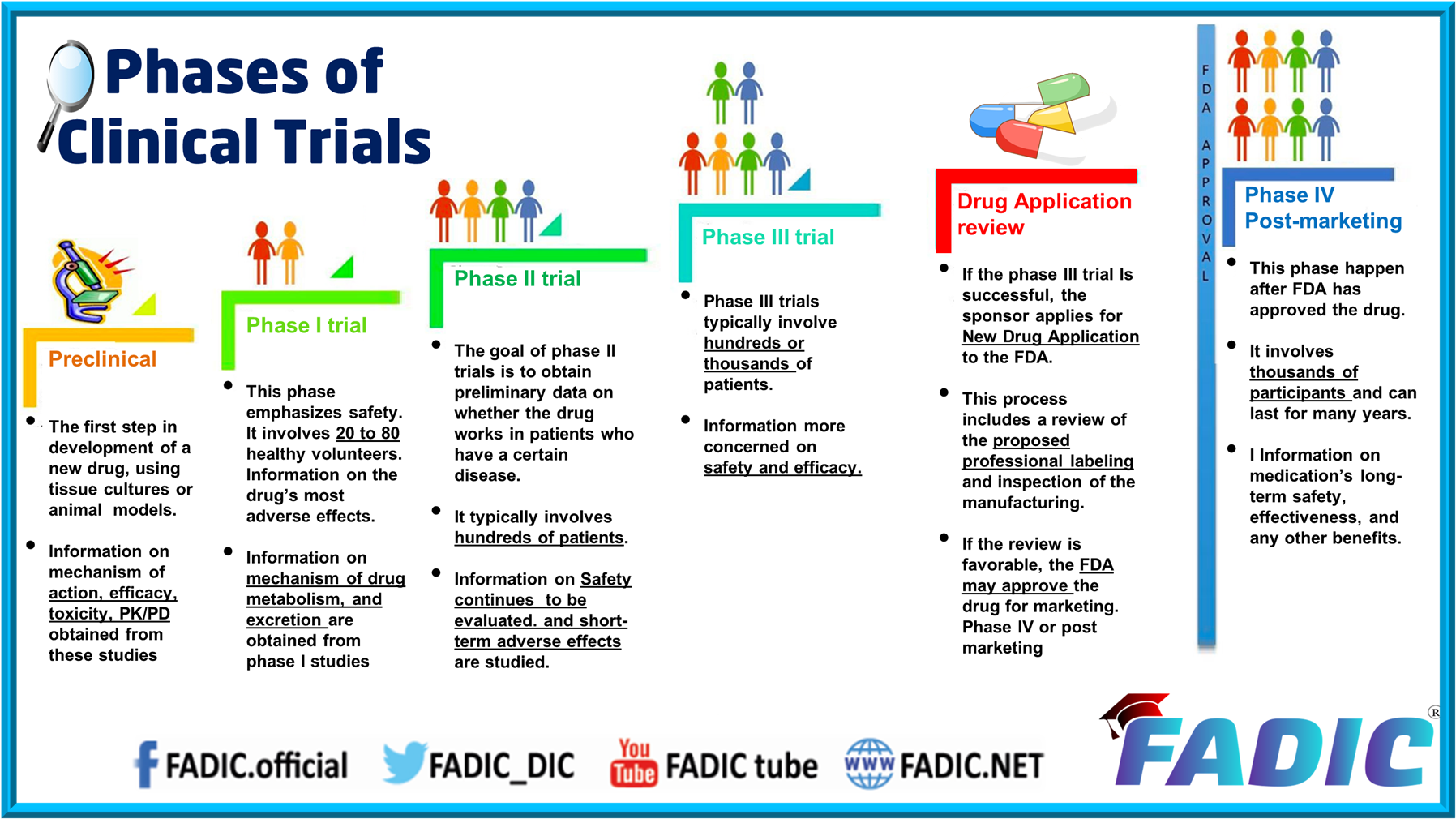
Phases Of Clinical Trials 30 Minutes E Course Learn about the design, purpose, and phases of clinical trials for drug development, from phase 1 to phase 4. find out how to submit an ind application to fda and get assistance from the review team. Clinical trials are divided into different phases. most trials are phase 1, 2 or 3. but some trials are classified as phase 0 and some as phase 4. Learn about the stages and goals of clinical trials for drug development, from preclinical testing to post marketing surveillance. find out the characteristics, participants, and success rates of each phase, as well as the ethical and regulatory aspects. Clinical trials are usually conducted in distinct phases. learn about each phase here.
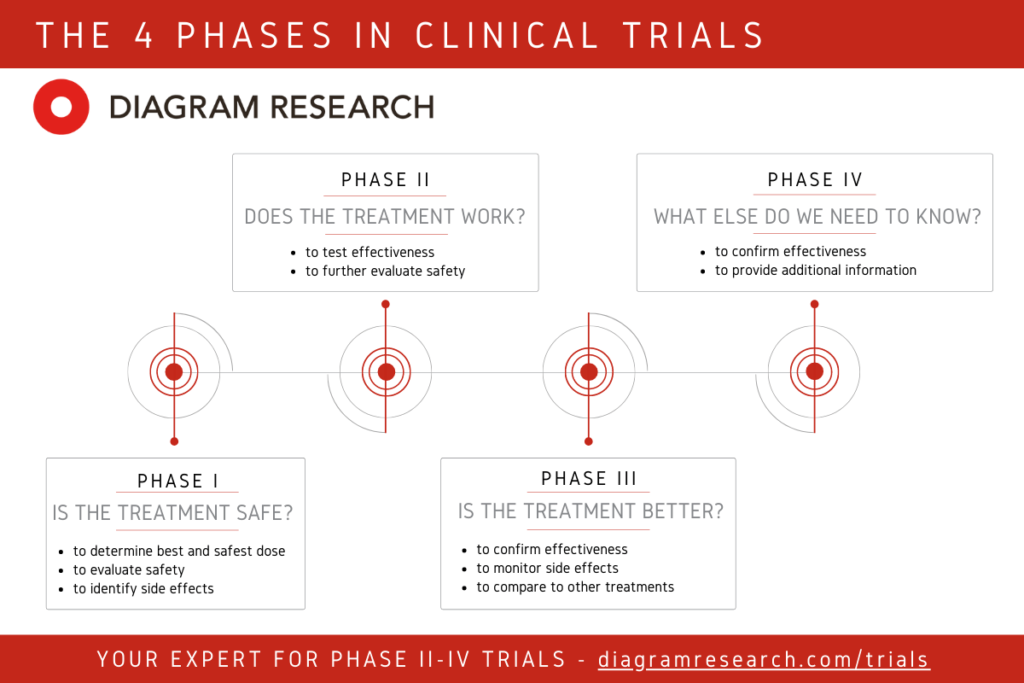
Phases In Clinical Trials Explained By Cro Diagram Research Learn about the stages and goals of clinical trials for drug development, from preclinical testing to post marketing surveillance. find out the characteristics, participants, and success rates of each phase, as well as the ethical and regulatory aspects. Clinical trials are usually conducted in distinct phases. learn about each phase here. Learn how clinical trials test new methods of diagnosing, treating, or preventing health conditions in several phases. find out what questions are asked, how participants are chosen, and what outcomes are measured in each phase. The 4 phases of clinical trials, from initial safety testing to post market monitoring. learn how each stage progresses, participant numbers, and objectives. After a phase i or ii trial, the researchers decide whether to move on to the next phase or to stop testing the treatment or procedure because it was unsafe or not effective. There are 4 clinical trials phases, each with its specific goal. phase 1 ensures safety, while phase 2 studies effectiveness. phase 3 confirms large scale success, and phase 4 monitors real world safety. every phase minimizes risks before new treatments reach patients.
Comments are closed.