Phases Of Clinical Development Eupati Toolbox
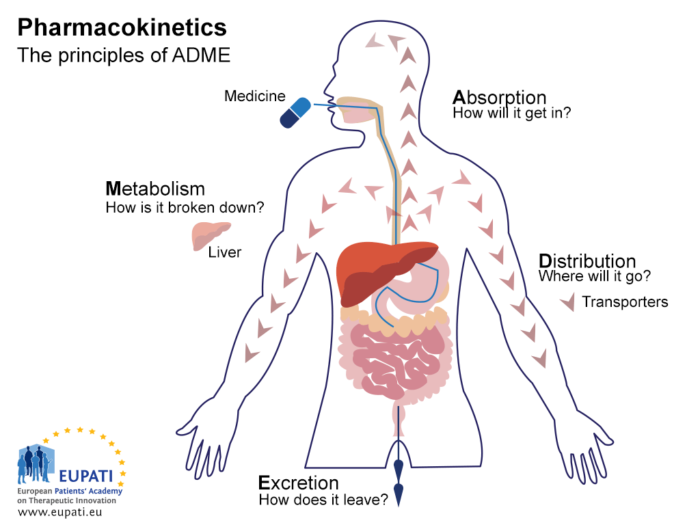
Pharmacokinetics Eupati Toolbox The diagram below explains the four phases of clinical development. the four phases of clinical development differ in terms of their objectives and features. a2 3.01 v1.1. Eupati has trained over 150 expert patients on medicines research & development – e.g. clinical trials, medicines regulations, health technology assessment. additionally, eupati offers and maintains the toolbox on medicines r&d, and coordinates a network of over 20 eupati national platforms.
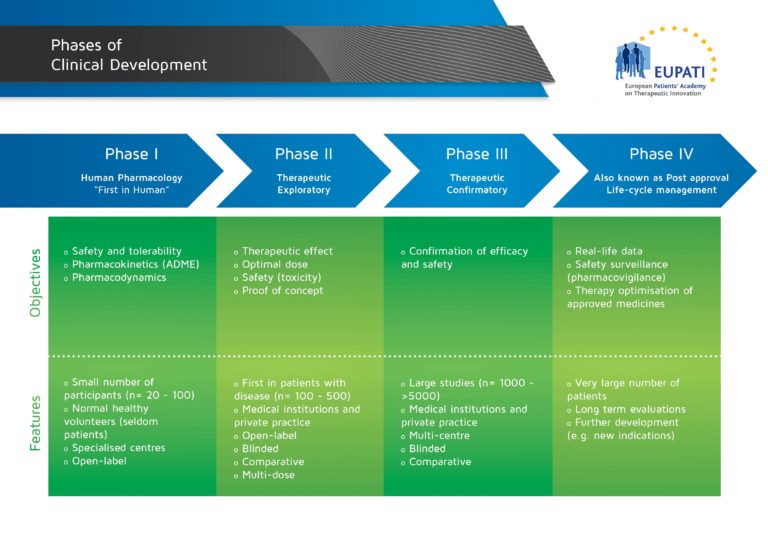
Basics Of Early Clinical Development Eupati Toolbox Designed for patients, patient groups, and anyone interested in learning more about medicines r&d, the freely accessible toolbox includes materials covering drug discovery, clinical development, regulatory affairs, medicines safety, pharmacovigilance, and health technology assessment. It describes the basic concepts of clinical trials as well as the types of studies in early clinical development, it also highlights the rights and obligations of trial participants, as well as the history of ethics in clinical research and the basic concepts of statistical methods. Many countries require an assessment of value, clinical effectiveness or cost effectiveness to decide on reimbursement. government or payers are increasingly supported through health technology assessment (hta) to take reimbursement decisions. The following are the guidance documents produced by eupati toolbox english.
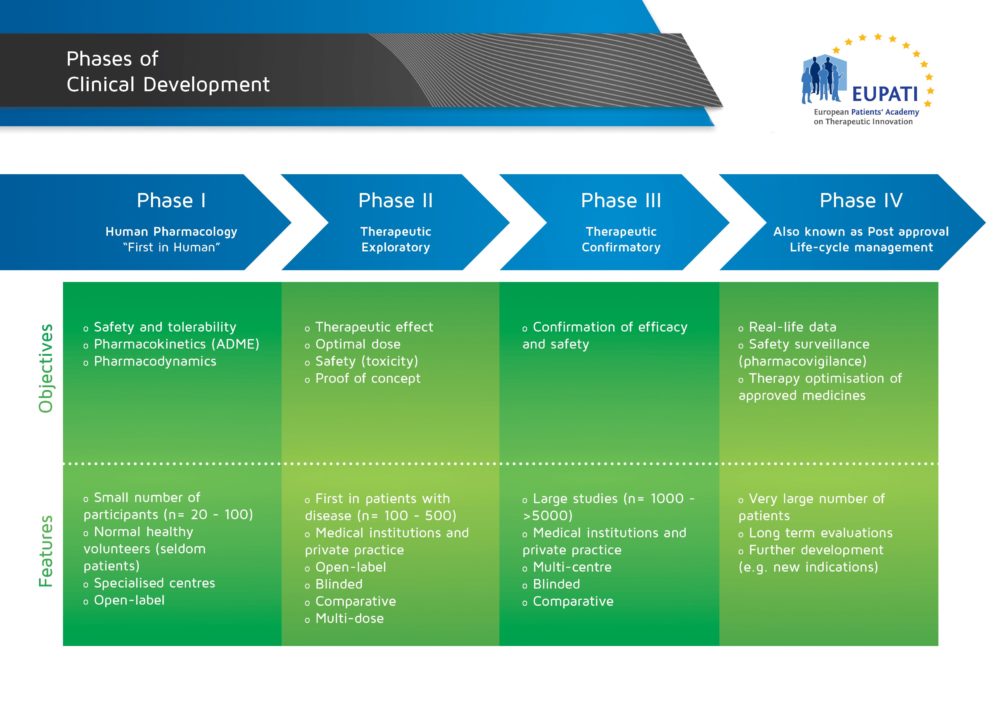
Basics Of Early Clinical Development Eupati Toolbox Many countries require an assessment of value, clinical effectiveness or cost effectiveness to decide on reimbursement. government or payers are increasingly supported through health technology assessment (hta) to take reimbursement decisions. The following are the guidance documents produced by eupati toolbox english. In order to achieve its goal of improving patient involvement (pi) in all processes of medicines r&d in switzerland and to obtain guidance and recommendations for future activities, eupati ch initiated a multi stakeholder survey on pi experiences, hurdles, and best practices. Content includes information rich modules and best in class educational materials on discovery research, clinical development, regulatory affairs, medicinal safety, pharmacovigilance and the principles of health technology assessment. Clinical studies are conducted in four steps, called ‘phases’ – each designed to answer separate research questions: the four phases of clinical development differ in terms of their objectives and features. Introduction to research methods and the conceptual description of trial phases.
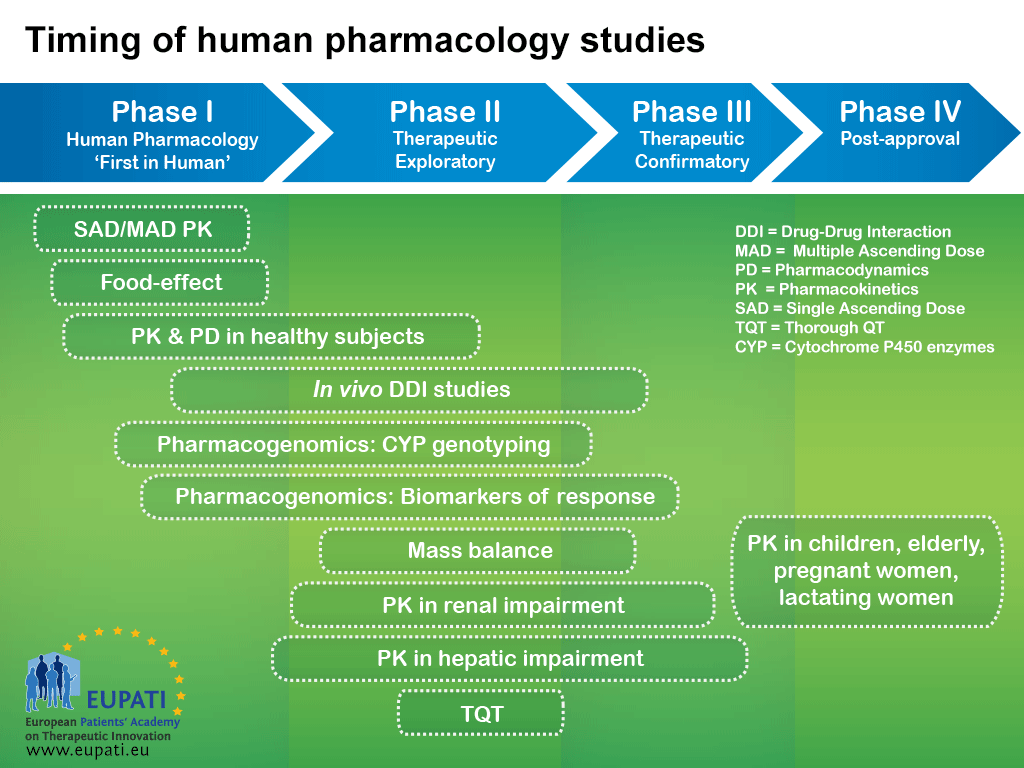
Basics Of Early Clinical Development Eupati Toolbox In order to achieve its goal of improving patient involvement (pi) in all processes of medicines r&d in switzerland and to obtain guidance and recommendations for future activities, eupati ch initiated a multi stakeholder survey on pi experiences, hurdles, and best practices. Content includes information rich modules and best in class educational materials on discovery research, clinical development, regulatory affairs, medicinal safety, pharmacovigilance and the principles of health technology assessment. Clinical studies are conducted in four steps, called ‘phases’ – each designed to answer separate research questions: the four phases of clinical development differ in terms of their objectives and features. Introduction to research methods and the conceptual description of trial phases.
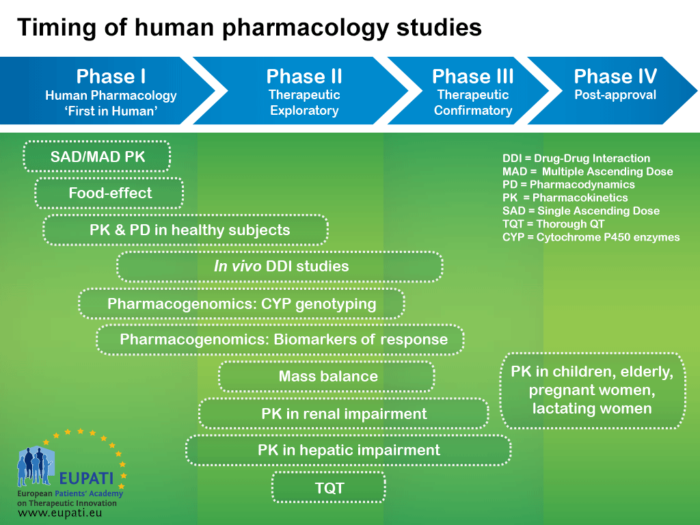
Basics Of Early Clinical Development Eupati Toolbox Clinical studies are conducted in four steps, called ‘phases’ – each designed to answer separate research questions: the four phases of clinical development differ in terms of their objectives and features. Introduction to research methods and the conceptual description of trial phases.
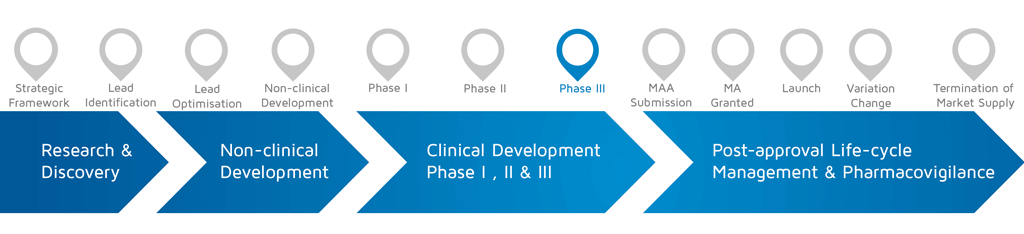
Patients Involved Clinical Trial Design Eupati Toolbox
Comments are closed.