Phases Of Clinical Development
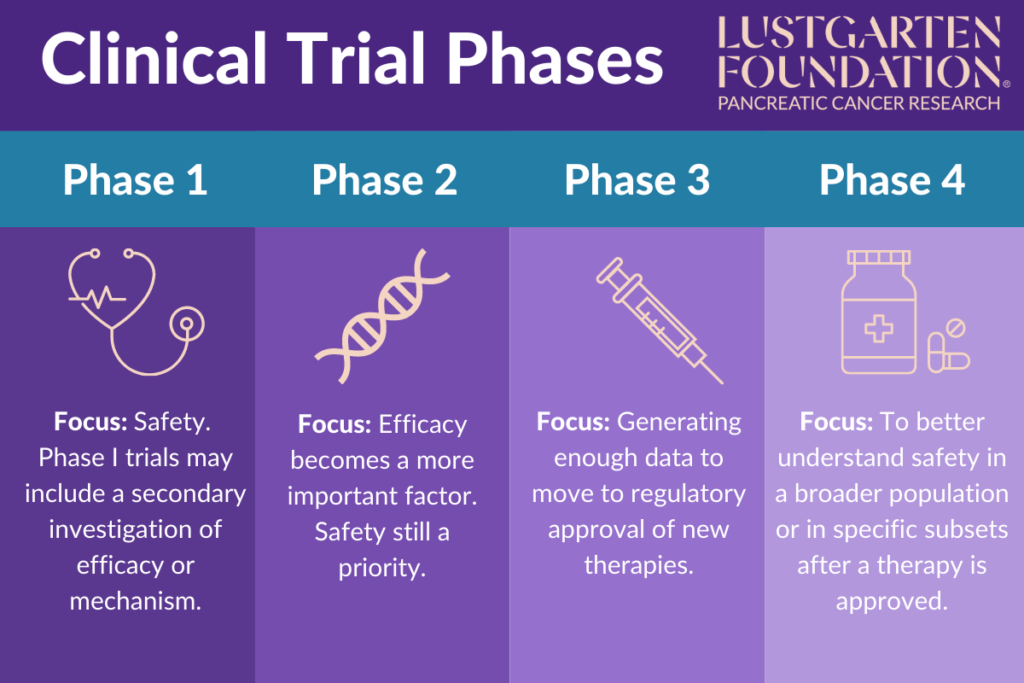
Clinical Trial Phases Instead, phase 2 studies provide researchers with additional safety data. researchers use these data to refine research questions, develop research methods, and design new phase 3 research. In clinical development, each phase, from preclinical to post marketing surveillance, carries its own set of challenges and opportunities. emphasizing these stages helps clarify the systematic approach required to bring new treatments to market safely and effectively.

Phases Of Clinical Trials Ncirs Clinical research is conducted on drug candidates, vaccine candidates, new medical devices, and new diagnostic assays. clinical trials testing potential medical products are commonly classified into four phases. the drug development process will normally proceed through all four phases over many years. [1] . The four phase clinical trial framework underpins modern drug development, balancing patient safety with society’s need for new therapies. each phase has distinct scientific aims and challenges, from the first dosing of humans in phase i to lifelong surveillance in phase iv. A new medication must pass through five distinct stages in order to be considered a “success”: 1) discovery and development; 2) preclinical research; 3) clinical trials; 4) fda drug review; and 5) fda post market safety monitoring. Explore clinical trial phases from preclinical studies to post marketing surveillance, and understand their role in drug development and approval.
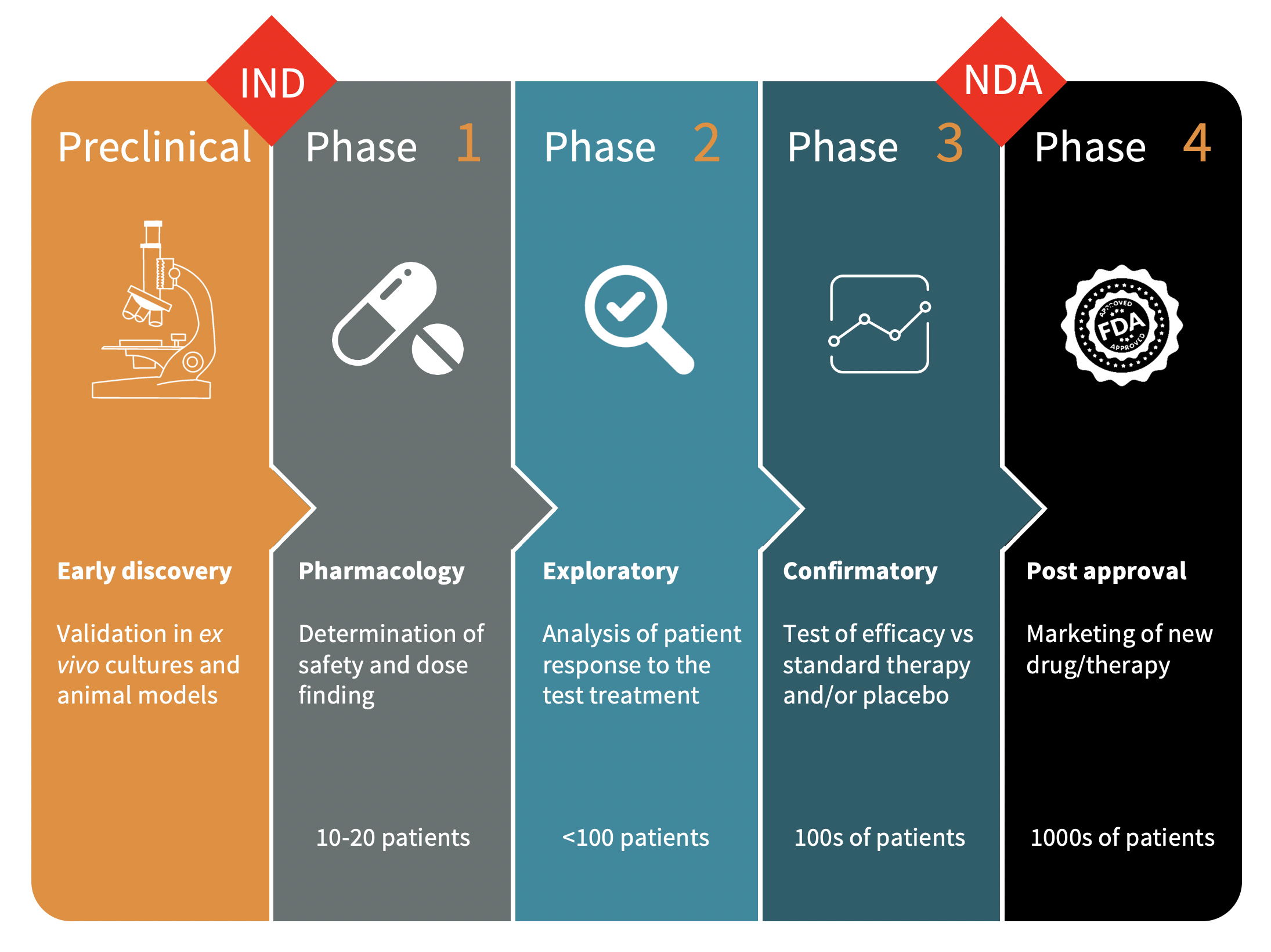
Phases Of Clinical Development A new medication must pass through five distinct stages in order to be considered a “success”: 1) discovery and development; 2) preclinical research; 3) clinical trials; 4) fda drug review; and 5) fda post market safety monitoring. Explore clinical trial phases from preclinical studies to post marketing surveillance, and understand their role in drug development and approval. The diagram below explains the four phases of clinical development. the four phases of clinical development differ in terms of their objectives and features. a2 3.01 v1.1. Therefore, in this review, we comprehensively describe the critical elements of clinical research that include clinical trial phases, types, and designs of clinical trials, operations of trial, audit, and management, and ethical concerns. This article outlines each phase in detail, examining how clinical trials evolve from microdosing studies to post marketing surveillance and how they align with regulatory expectations across the drug development lifecycle. The clinical development process consists of four sequential phases, each characterized by distinct objectives, study designs, and participant criteria. phase i trials primarily evaluate safety profiles and determine appropriate dosing parameters in small participant groups.
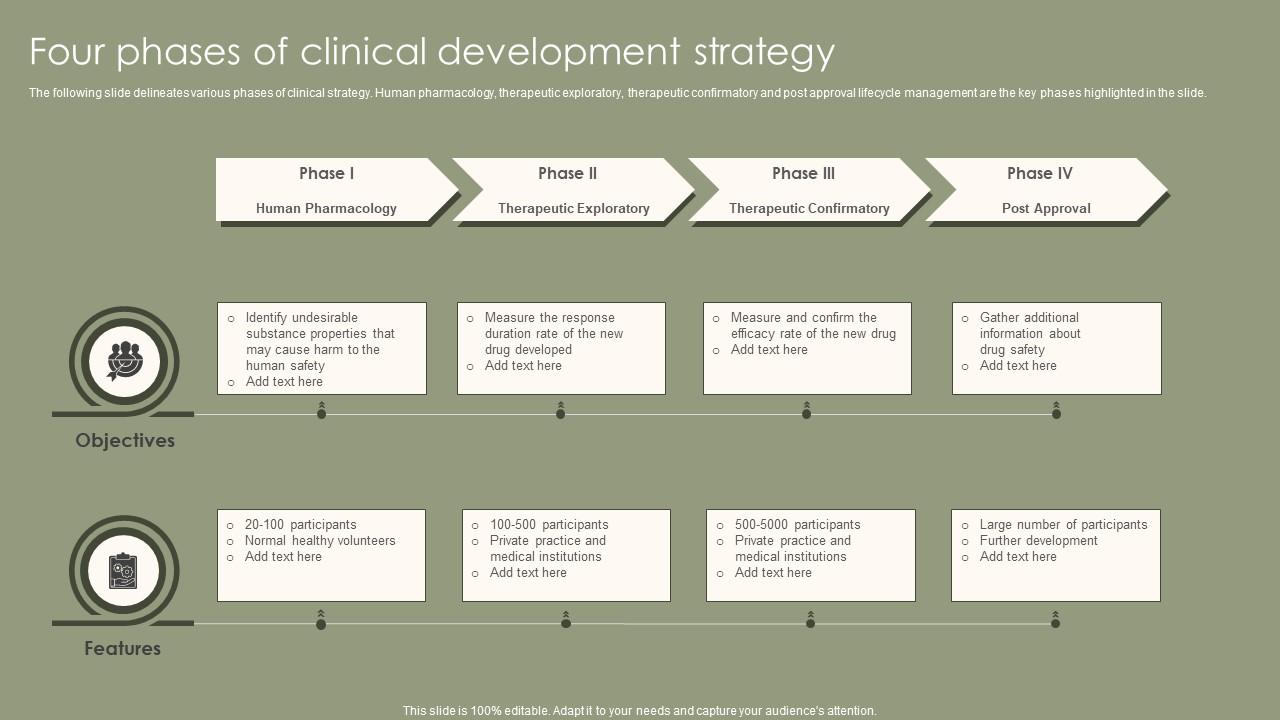
Phases Of Clinical Development The diagram below explains the four phases of clinical development. the four phases of clinical development differ in terms of their objectives and features. a2 3.01 v1.1. Therefore, in this review, we comprehensively describe the critical elements of clinical research that include clinical trial phases, types, and designs of clinical trials, operations of trial, audit, and management, and ethical concerns. This article outlines each phase in detail, examining how clinical trials evolve from microdosing studies to post marketing surveillance and how they align with regulatory expectations across the drug development lifecycle. The clinical development process consists of four sequential phases, each characterized by distinct objectives, study designs, and participant criteria. phase i trials primarily evaluate safety profiles and determine appropriate dosing parameters in small participant groups.

Clinical Trial Phases With Development And Product Surveillance Medical Res This article outlines each phase in detail, examining how clinical trials evolve from microdosing studies to post marketing surveillance and how they align with regulatory expectations across the drug development lifecycle. The clinical development process consists of four sequential phases, each characterized by distinct objectives, study designs, and participant criteria. phase i trials primarily evaluate safety profiles and determine appropriate dosing parameters in small participant groups.

Clinical Trial Phases With Development And Product Surveillance
Comments are closed.