Phases In Clinical Trials Explained By Cro Diagram Research
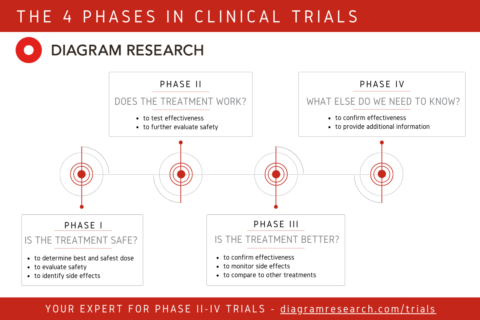
Phases In Clinical Trials Explained By Cro Diagram Research Clear and simple explanation of the phases of clinical trials, both for ip (investigational product) trials and md (medical device) trials. Explore the purpose, process, and characteristics of the four phases of clinical research, crucial for validating drug safety and efficacy.

Phases In Clinical Trials Explained By Cro Diagram Research In this blog, we’ll break down the key stages of the clinical trial workflow and discuss how clinical research organizations (cros) help navigate this complex journey. The development of new drugs and therapies involves a meticulous process called clinical trials. these trials are conducted in stages, with each phase serving a specific purpose. there are four main 4 phases of clinical trials: phase i, phase ii, phase iii, and phase iv. Clinical trials are the scientific cornerstone of modern medicine, providing the evidence base for the safety and efficacy of new drugs and therapies. they are traditionally organized into four successive phases – i, ii, iii, and iv – each with distinct objectives, designs, and regulatory roles. This comprehensive guide explains how clinical trials are conducted step by step, and the critical role clinical research organizations (cros) play in delivering successful outcomes.

4 Phases Of Clinical Trials Lambda Cro Clinical Research Clinical trials are the scientific cornerstone of modern medicine, providing the evidence base for the safety and efficacy of new drugs and therapies. they are traditionally organized into four successive phases – i, ii, iii, and iv – each with distinct objectives, designs, and regulatory roles. This comprehensive guide explains how clinical trials are conducted step by step, and the critical role clinical research organizations (cros) play in delivering successful outcomes. Clinical research is conducted on drug candidates, vaccine candidates, new medical devices, and new diagnostic assays. Therefore, in this review, we comprehensively describe the critical elements of clinical research that include clinical trial phases, types, and designs of clinical trials, operations of trial, audit, and management, and ethical concerns. The phases of clinical trial research. clinical trials pass through a series of phases as the trial sponsor gains more evidence around the investigational drug or biologic. Below is the 4 phases of clinical trials, presented with clarity to highlight their critical role in shaping successful and impactful therapeutic development.
Comments are closed.