Phase Ii Clinical Trials

Phase Ii Clinical Trials A Critical Step In The Evaluation Of New Phase ii trials are designed to determine if the new treatment has sufficiently promising efficacy to warrant further investigation in a large scale randomized phase iii trial, as well as to further assess safety. these studies usually involve a few hundred patients. Explore phase ii clinical trials: understand goals, real world examples, and insights that reshape drug development and regulatory pathways.
Phase Ii Clinical Trials A Critical Step In The Evaluation Of New Most drugs undergoing phase iii clinical trials can be marketed under fda norms with proper recommendations and guidelines through a new drug application (nda) containing all manufacturing, preclinical, and clinical data. Phase ii studies can be broken down into phase iia and phase iib, where phase iia is a pilot clinical trial to evaluate effectiveness and safety in selected populations of patients and phase iib studies are well controlled trials to evaluate efficacy and safety in patients. Much of the attrition occurs in phase ii: many candidate drugs that show safety in early trials fail to demonstrate adequate efficacy or acceptable safety in patients. these statistics underscore the challenge of translating preclinical promise into clinical benefit. Phase 2 trials shift focus from initial safety (phase 1’s primary aim) to evaluating a treatment’s ability to affect the target disease. this stage provides the first clear signal of whether the drug demonstrates biological activity in people who have the condition it is intended to treat.

Phase Ii Clinical Trials A Critical Step In The Evaluation Of New Much of the attrition occurs in phase ii: many candidate drugs that show safety in early trials fail to demonstrate adequate efficacy or acceptable safety in patients. these statistics underscore the challenge of translating preclinical promise into clinical benefit. Phase 2 trials shift focus from initial safety (phase 1’s primary aim) to evaluating a treatment’s ability to affect the target disease. this stage provides the first clear signal of whether the drug demonstrates biological activity in people who have the condition it is intended to treat. Phase 2 trials look at how well the treatment works and monitors its safety. during this phase, researchers get information on how the treatment works and whether it has side effects. Phase 2 acts as a bridge to large scale phase 3 trials, which determine the drug’s final outcome. this chapter provides an overview of phase 2 clinical trials. phase 2 clinical trials, or “therapeutic exploratory trials,” assess a treatment’s efficacy and safety in several hundred participants. Phase ii research is normally randomised and controlled. the trials typically include between 100 300 participants with the disease or condition of interest. typically, this phase spans anywhere from a few months to a few years. the trial can be subdivided into 2 parts: phase iia and phase iib. Following preclinical and first in human (phase i) studies, an investigational drug or device is studied in subjects who have the disorder of interest. this phase of development is commonly called phase ii or exploratory therapeutic development.
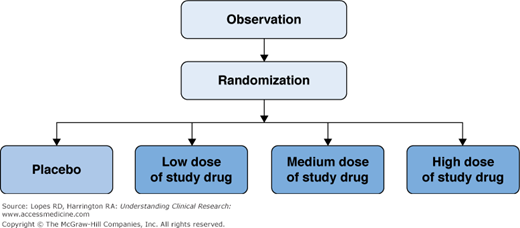
Phase Ii Clinical Trials Basicmedical Key Phase 2 trials look at how well the treatment works and monitors its safety. during this phase, researchers get information on how the treatment works and whether it has side effects. Phase 2 acts as a bridge to large scale phase 3 trials, which determine the drug’s final outcome. this chapter provides an overview of phase 2 clinical trials. phase 2 clinical trials, or “therapeutic exploratory trials,” assess a treatment’s efficacy and safety in several hundred participants. Phase ii research is normally randomised and controlled. the trials typically include between 100 300 participants with the disease or condition of interest. typically, this phase spans anywhere from a few months to a few years. the trial can be subdivided into 2 parts: phase iia and phase iib. Following preclinical and first in human (phase i) studies, an investigational drug or device is studied in subjects who have the disorder of interest. this phase of development is commonly called phase ii or exploratory therapeutic development.

Ppt Phase Ii Clinical Trials Powerpoint Presentation Free Download Phase ii research is normally randomised and controlled. the trials typically include between 100 300 participants with the disease or condition of interest. typically, this phase spans anywhere from a few months to a few years. the trial can be subdivided into 2 parts: phase iia and phase iib. Following preclinical and first in human (phase i) studies, an investigational drug or device is studied in subjects who have the disorder of interest. this phase of development is commonly called phase ii or exploratory therapeutic development.
Comments are closed.