Phase I Clinical Trials Explained Objectives Risks Process
Phase I Clinical Trials Explained Objectives Risks Process Phase i clinical trials mark the critical first step in evaluating a new drug’s safety and tolerability in humans. these trials set the groundwork for all subsequent phases, determining whether a compound has the potential to advance toward broader patient populations. Injecting new drugs first into healthy volunteers (phase i) ties to minimal risk principles, whereas later trials in patients (phase ii iii) justify higher risk by potential benefit.

Phase I Clinical Trials Explained Objectives Risks Process Learn about phase i clinical trials, the initial stage focusing on a new treatment's safety, tolerability, and how it's processed by the body (pharmacokinetics pharmacodynamics). discover the importance of "first administration to humans," bioequivalence studies, and dose escalation. This guide outlines the core components of a phase 1 clinical trial, including the key objectives, participant selection process, dose escalation strategy, and intense safety monitoring. Explore phase i clinical trials focused on safety and dosage, covering objectives, design strategies, regulatory pathways, and best practices for early human testing. Phase 1 studies are closely monitored and gather information about how a drug interacts with the human body. researchers adjust dosing schemes based on animal data to find out how much of a.

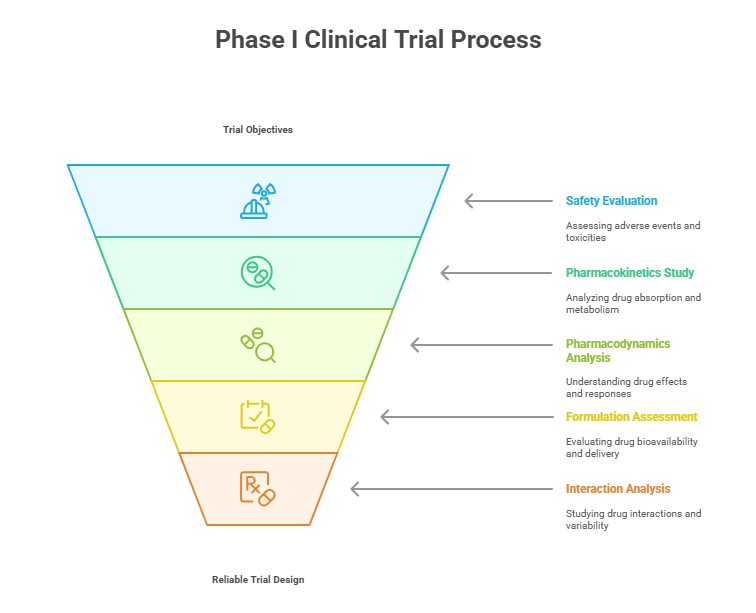
Phase I Clinical Trials Explained Objectives Risks Process Explore phase i clinical trials focused on safety and dosage, covering objectives, design strategies, regulatory pathways, and best practices for early human testing. Phase 1 studies are closely monitored and gather information about how a drug interacts with the human body. researchers adjust dosing schemes based on animal data to find out how much of a. Phase i clinical trials primarily aim to address two key questions: 1. what adverse reactions may the drug cause? 2. how is the drug absorbed, distributed, metabolized, and excreted in humans? to comprehensively answer these questions, a series of critical investigations must be completed. A phase 1 clinical trial is the first time an experimental drug or treatment is tested in humans. its primary goal is safety: researchers want to find out whether a new compound is safe enough to keep testing, what side effects it causes, and how the human body processes it. Phase 1 trials primarily investigate the pk pd, safety, dosing, and toxicities of a compound drug. while completing the previous phase is generally necessary to proceed to the next, phase 1 studies can be conducted alongside efficacy clinical trials. Clinical trials progress through phases i to iv, each focusing on safety, effectiveness, comparison, and post market monitoring. phase i trials primarily test safety and dosage in a small group of participants.

Phase I Clinical Trials Explained Objectives Risks Process Phase i clinical trials primarily aim to address two key questions: 1. what adverse reactions may the drug cause? 2. how is the drug absorbed, distributed, metabolized, and excreted in humans? to comprehensively answer these questions, a series of critical investigations must be completed. A phase 1 clinical trial is the first time an experimental drug or treatment is tested in humans. its primary goal is safety: researchers want to find out whether a new compound is safe enough to keep testing, what side effects it causes, and how the human body processes it. Phase 1 trials primarily investigate the pk pd, safety, dosing, and toxicities of a compound drug. while completing the previous phase is generally necessary to proceed to the next, phase 1 studies can be conducted alongside efficacy clinical trials. Clinical trials progress through phases i to iv, each focusing on safety, effectiveness, comparison, and post market monitoring. phase i trials primarily test safety and dosage in a small group of participants.
Comments are closed.