Phase Diagrams
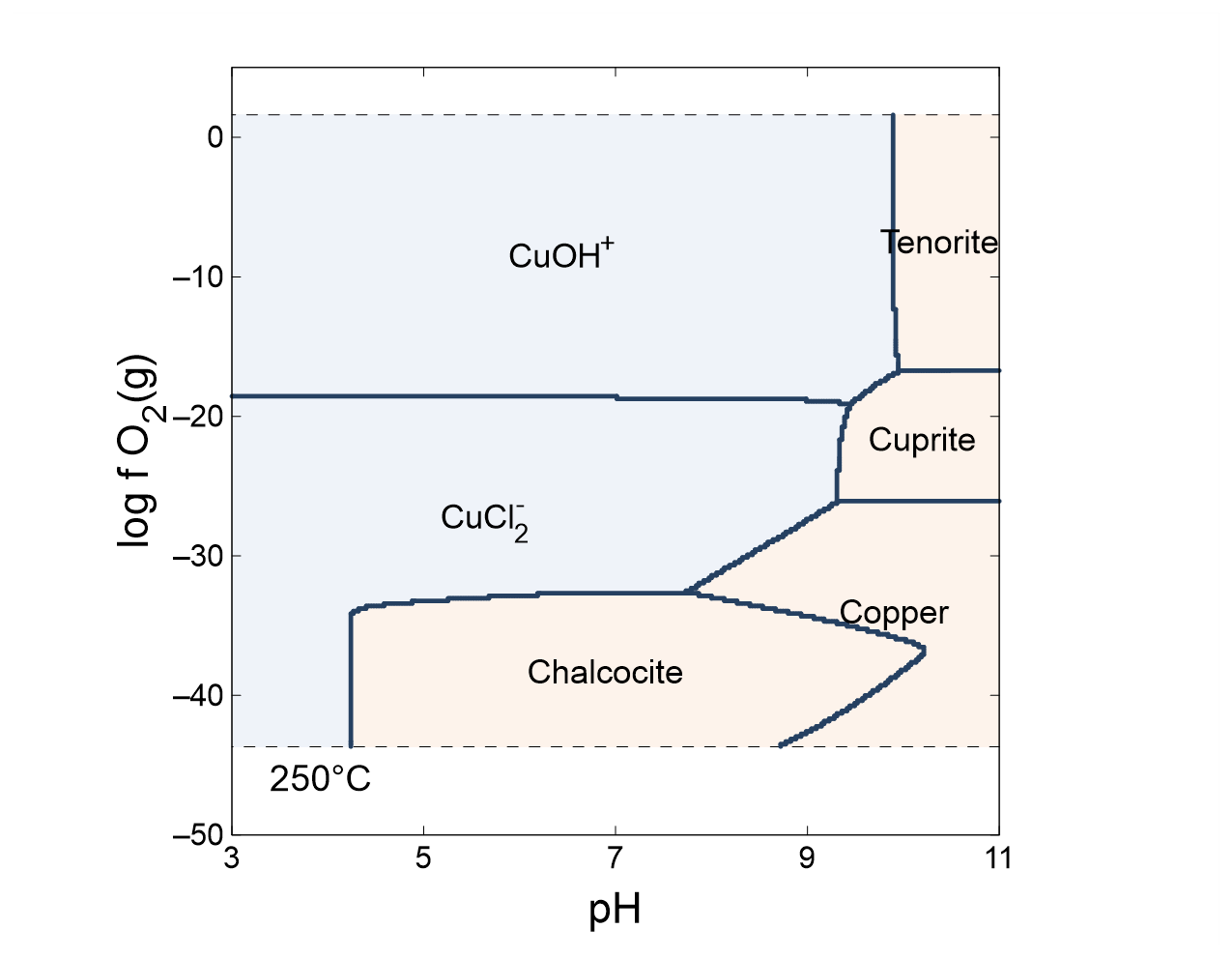
The Geochemist S Workbench Online Academy Phase Diagrams A phase diagram in physical chemistry, engineering, mineralogy, and materials science is a type of chart used to show conditions (pressure, temperature, etc.) at which thermodynamically distinct phases (such as solid, liquid or gaseous states) occur and coexist at equilibrium. Learn about phase diagrams, graphs that show the physical states of a substance under different conditions of temperature and pressure. explore the types of phase transitions, such as melting, boiling, sublimation, and vaporization, and their curves on the phase diagram.

Understanding Ternary Phase Diagrams A Guide To Multicomponent Systems Learn what a phase diagram is and how to read it. see examples of phase diagrams of water and carbon dioxide, and understand the concepts of triple point and critical point. A phase diagram shows a material's stable phases at different levels of temperature and pressure, enabling the prediction of melting, boiling, or crystallization points. Key points on a phase diagram include the triple point, critical point, fusion curve (melting), vaporization curve, and sublimation curve, each representing specific phase transitions. in this article, we will look into what a phase diagram is, its definitions, components, types, applications, etc., in detail. Phase diagrams are graphical representations that illustrate the various phases of a substance as a function of temperature and composition, showing the conditions under which distinct phases, such as solid, liquid, and gas, exist and coexist.
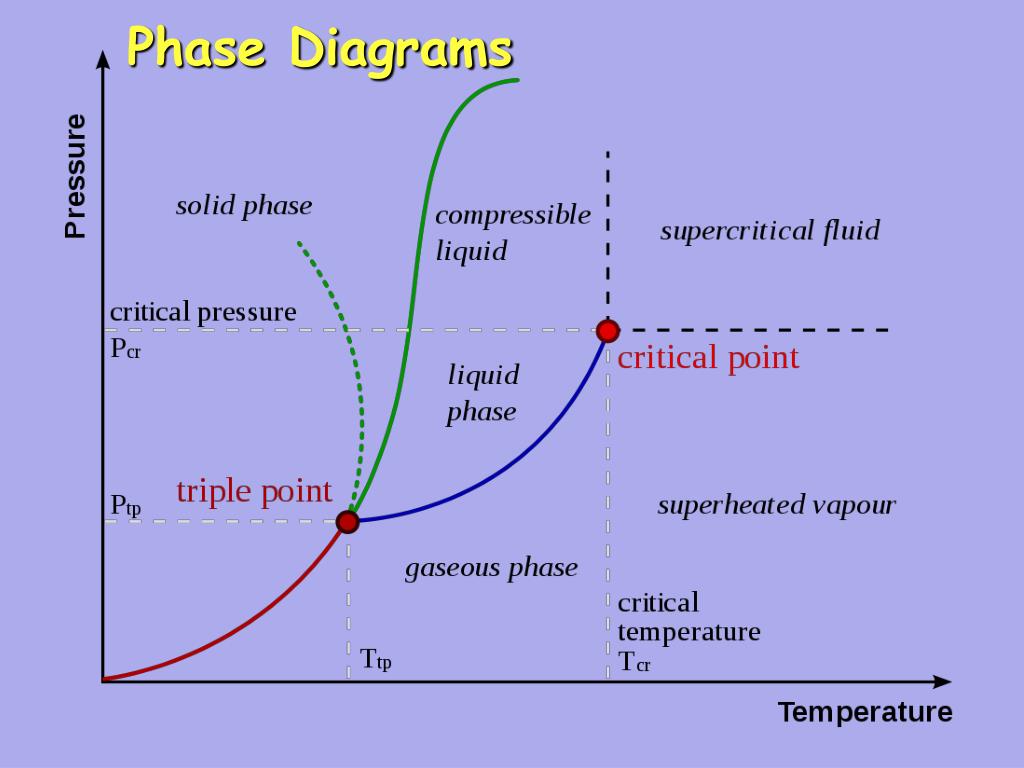
Ppt Phase Diagrams Powerpoint Presentation Free Download Id 704549 Key points on a phase diagram include the triple point, critical point, fusion curve (melting), vaporization curve, and sublimation curve, each representing specific phase transitions. in this article, we will look into what a phase diagram is, its definitions, components, types, applications, etc., in detail. Phase diagrams are graphical representations that illustrate the various phases of a substance as a function of temperature and composition, showing the conditions under which distinct phases, such as solid, liquid, and gas, exist and coexist. A phase diagram lets you work out exactly what phases are present at any given temperature and pressure. in the cases we'll be looking at on this page, the phases will simply be the solid, liquid or vapour (gas) states of a pure substance. Phase diagrams are plots that show the different phases of a substance across multiple variables, such as temperature and pressure. learn about the features, types, and examples of phase diagrams, and how they relate to states of matter, intermolecular forces, and physical properties. Phase diagrams are combined plots of three pressure temperature equilibrium curves: solid liquid, liquid gas, and solid gas. these curves represent the relationships between phase transition temperatures and pressures. Learn about phase diagrams, the equilibrium conditions between different phases of materials, and how to construct them. see examples of phase diagrams for water, iron, copper nickel, and lead tin systems.
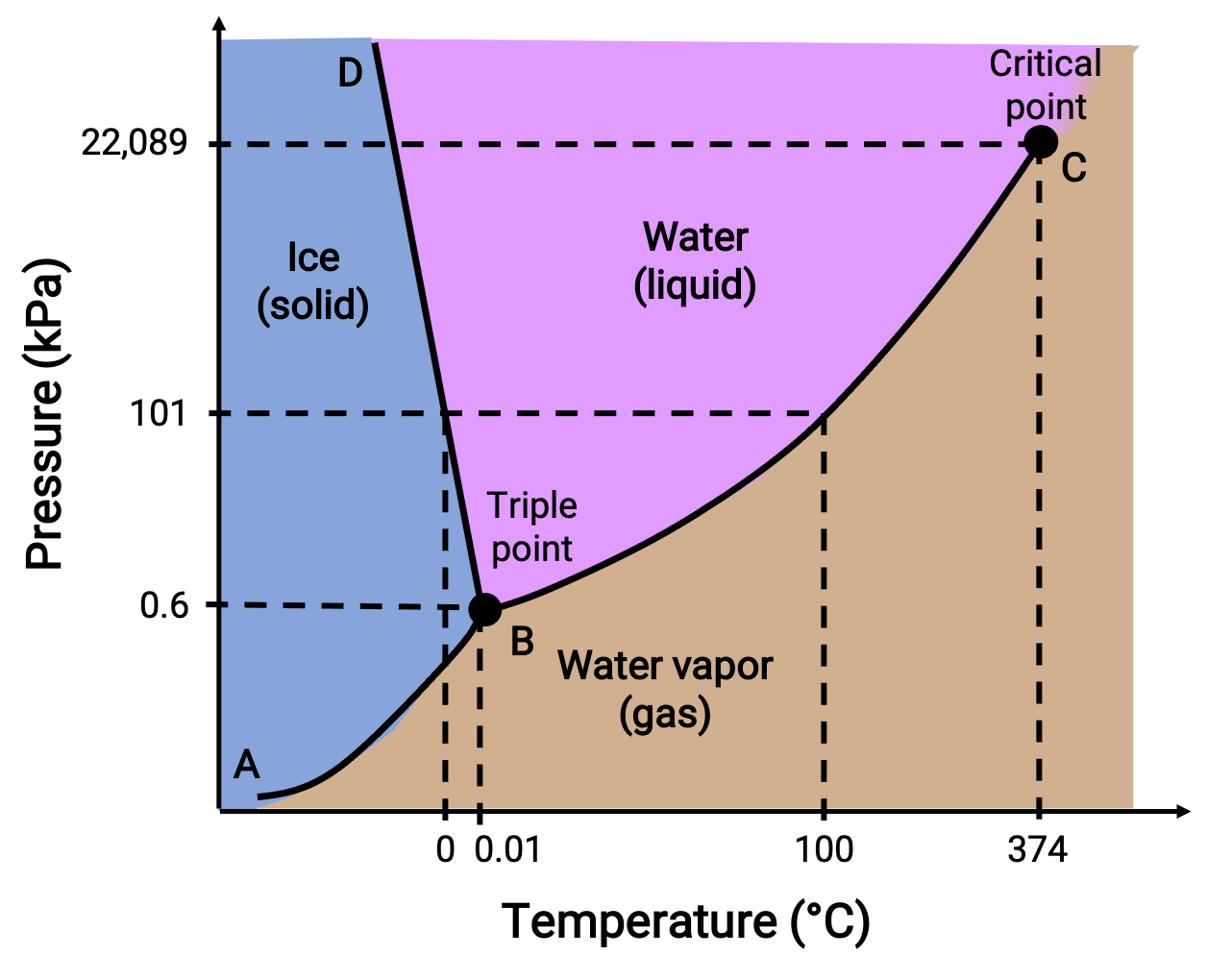
Video Phase Diagrams A phase diagram lets you work out exactly what phases are present at any given temperature and pressure. in the cases we'll be looking at on this page, the phases will simply be the solid, liquid or vapour (gas) states of a pure substance. Phase diagrams are plots that show the different phases of a substance across multiple variables, such as temperature and pressure. learn about the features, types, and examples of phase diagrams, and how they relate to states of matter, intermolecular forces, and physical properties. Phase diagrams are combined plots of three pressure temperature equilibrium curves: solid liquid, liquid gas, and solid gas. these curves represent the relationships between phase transition temperatures and pressures. Learn about phase diagrams, the equilibrium conditions between different phases of materials, and how to construct them. see examples of phase diagrams for water, iron, copper nickel, and lead tin systems.
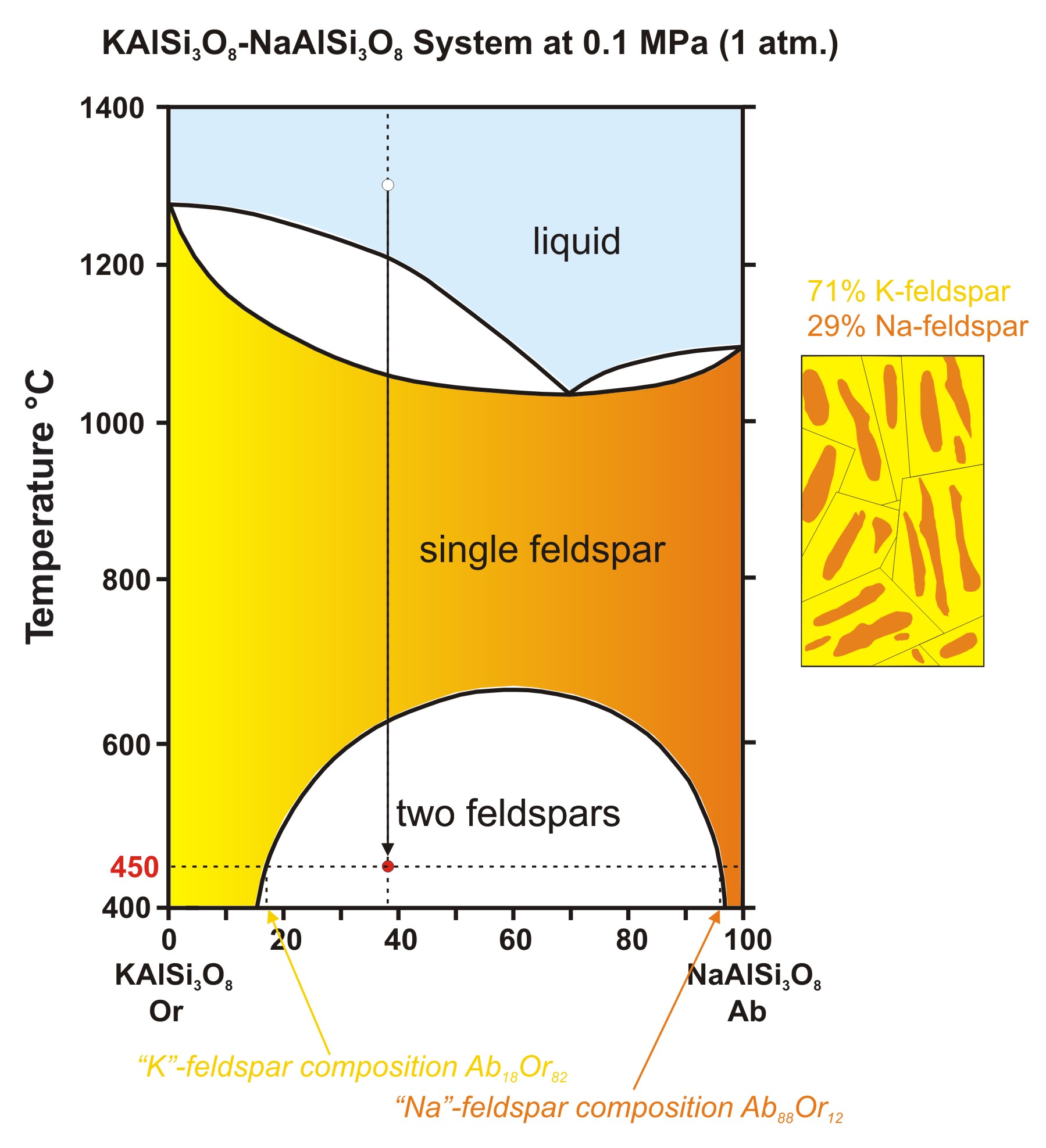
Phase Diagrams And Pseudosections Phase diagrams are combined plots of three pressure temperature equilibrium curves: solid liquid, liquid gas, and solid gas. these curves represent the relationships between phase transition temperatures and pressures. Learn about phase diagrams, the equilibrium conditions between different phases of materials, and how to construct them. see examples of phase diagrams for water, iron, copper nickel, and lead tin systems.
Comments are closed.