Phase Changes Physics

Thermal Physics Phase Changes 1 2 Teaching Resources Phase transitions play an important theoretical and practical role in the study of heat flow. in melting (or “ fusion ”), a solid turns into a liquid; the opposite process is freezing. in evaporation, a liquid turns into a gas; the opposite process is condensation. A phase change or phase transition is a physical transition between solid, liquid, gaseous, and sometimes plasma states of matter, due to changes in temperature or pressure.

Let S Talk Vocab Physics Changes Of Phase Thermodynamics In physics, chemistry and biology, a phase transition (or phase change) is the physical process of transition between one state of a medium and another. commonly the term is used to refer to changes among the basic states of matter: solid, liquid, and gas, and in rare cases, plasma. This chapter expands on the topics of heat and thermodynamics by discussing the states of matter and how substances change their phase. we’ll learn about the energy required to create phase changes, and analyze the heat, temperature, and phase diagrams of different substances. Phase changes among the various phases of matter depend on temperature and pressure. the existence of the three phases with respect to pressure and temperature can be described in a phase diagram. Comparing both processes we can derive the following definition: a phase change (pc) is a change in the arrangement of atoms, molecules or particles of a substance of fixed chemical composition that is induced from the outside and will manifest itself by the presence of new properties of matter.

Phase Change Learn Its Meaning Examples Characteristics Phase changes among the various phases of matter depend on temperature and pressure. the existence of the three phases with respect to pressure and temperature can be described in a phase diagram. Comparing both processes we can derive the following definition: a phase change (pc) is a change in the arrangement of atoms, molecules or particles of a substance of fixed chemical composition that is induced from the outside and will manifest itself by the presence of new properties of matter. A phase change is when matter changes to from one state (solid, liquid, gas, plasma) to another. (see figure 1). these changes occur when sufficient energy is supplied to the system (or a sufficient amount is lost), and also occur when the pressure on the system is changed. Transitions between solid, liquid, and gaseous phases typically involve large amounts of energy compared to the specific heat. Phase transitions play an important theoretical and practical role in the study of heat flow. in melting (or “fusion”), a solid turns into a liquid; the opposite process is freezing. in evaporation, a liquid turns into a gas; the opposite process is condensation. Phase changes are physical changes in which a substance shifts from one state of matter to another, like ice melting into water or water boiling into steam. these transformations depend on two factors: particle attractions and kinetic energy.

Change Of Phase State Phase Transition Physics Tutorials A phase change is when matter changes to from one state (solid, liquid, gas, plasma) to another. (see figure 1). these changes occur when sufficient energy is supplied to the system (or a sufficient amount is lost), and also occur when the pressure on the system is changed. Transitions between solid, liquid, and gaseous phases typically involve large amounts of energy compared to the specific heat. Phase transitions play an important theoretical and practical role in the study of heat flow. in melting (or “fusion”), a solid turns into a liquid; the opposite process is freezing. in evaporation, a liquid turns into a gas; the opposite process is condensation. Phase changes are physical changes in which a substance shifts from one state of matter to another, like ice melting into water or water boiling into steam. these transformations depend on two factors: particle attractions and kinetic energy.
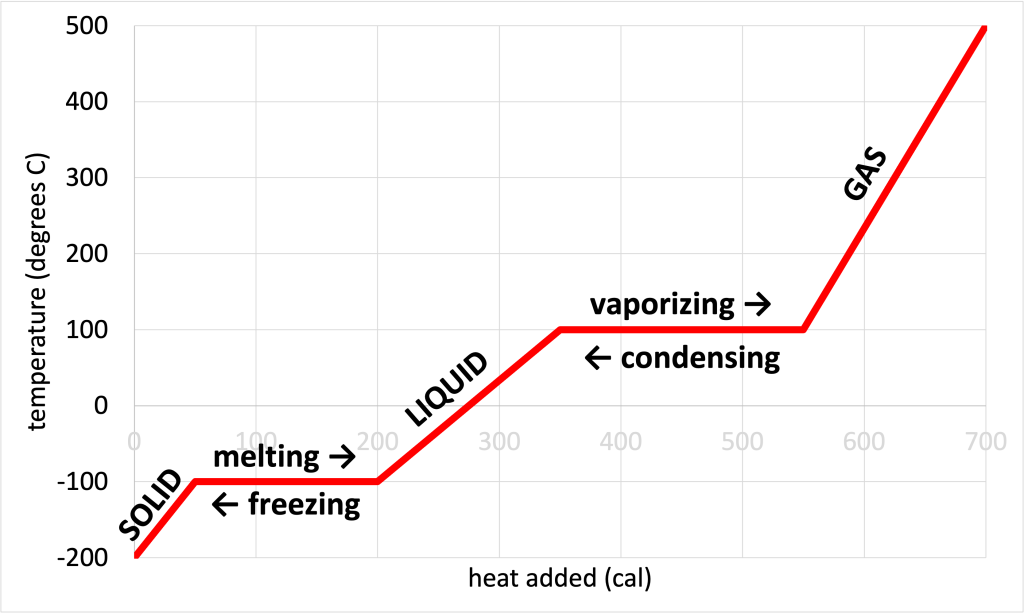
17 Phase Change Conceptual Physics Phase transitions play an important theoretical and practical role in the study of heat flow. in melting (or “fusion”), a solid turns into a liquid; the opposite process is freezing. in evaporation, a liquid turns into a gas; the opposite process is condensation. Phase changes are physical changes in which a substance shifts from one state of matter to another, like ice melting into water or water boiling into steam. these transformations depend on two factors: particle attractions and kinetic energy.
Comments are closed.