Pharmacokinetics Eupati Toolbox

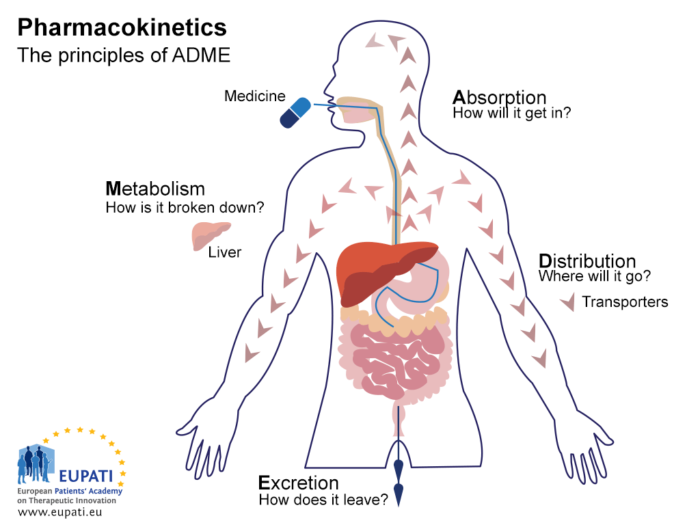
Safety Communication Eupati Toolbox Pdf Pharmacovigilance Pharmacokinetics is the study of what the body does to medicine. it studies the absorption, distribution, metabolism, and excretion of the medicine (adme), as well as bioavailability. Just like a real toolbox that any carpenter or electrician uses, the eupati toolbox contains all the ‘tools’ you need around medicines research and development and patient engagement.
Pharmacokinetics Eupati Toolbox Below you will find some of eupati's resources available for patients and researchers interested in ppi in medicines development and beyond. this elearning platform offers a learning catalogue that can be taken online and on demand at your own pace and preferred order. Eupati has trained over 150 expert patients on medicines research & development – e.g. clinical trials, medicines regulations, health technology assessment. additionally, eupati offers and maintains the toolbox on medicines r&d, and coordinates a network of over 20 eupati national platforms. There are two key areas of pharmacology: a rigorous non clinical programme must be completed before a medicine may be tested in humans for the first time in phase i clincial trials. the animal models used in non clinical trials are carefully selected, taking various factors into consideration. The purpose of the toolbox is to provide access to well structured, comprehensive, scientifically reliable, and user friendly educational materials for patients on a variety of topics in these areas.

Eupati Toolbox Eupati Toolbox There are two key areas of pharmacology: a rigorous non clinical programme must be completed before a medicine may be tested in humans for the first time in phase i clincial trials. the animal models used in non clinical trials are carefully selected, taking various factors into consideration. The purpose of the toolbox is to provide access to well structured, comprehensive, scientifically reliable, and user friendly educational materials for patients on a variety of topics in these areas. Establish the necessary infrastructure and govern ance to ensure eupati’s continuous sustainability on the european, national and global level in the mid and long term. The process by which a medicine is distributed from one location to another within the body. see also pharmacokinetics. the key principles of pharmacokinetics – the study of the effect the body has on a medicine – are represented in the acronym adme. Eupati is enhancing patient involvement by providing information, education, and opportunities for collaboration for patients and researchers throughout all aspects of the health innovation ecosystem. Data gathered during pharmacokinetic studies provide information about what happens to a medicine in the body over time. scientific and mathematical models based on this information help to understand and predict the journey of the medicine and its metabolites through the body.
Comments are closed.