Pharmaceutical Validation Pptx

Dokumen Tips Validation Pharma Industry Pharmaceutics The document provides a comprehensive overview of validation in the pharmaceutical industry, detailing its importance, types, and guidelines set forth by regulatory bodies like the fda and ich. Finally it can be concluded that process validation is a key element in the quality assurance of pharmaceutical product as the end product testing is not sufficient to assure the quality of finished product.

Pharmaceutical Validation Pptx Validation is an integral part of quality assurance; it involves the systematic study of systems, facilities and processes aimed at determining whether they perform their intended functions adequately and consistently as specified. This approach is undertaken whenever the process for a new formula (or within a new facility) must be validated before routine pharmaceutical production commences. Finalization of the validation process: a validation report must be prepared at the conclusion of validation activities. it should summarize and reference all protocols and results. obviously, a conclusion is required: “is the process valid”, but strangely often omitted. Validation in the pharmaceutical industry is a systematic approach to ensuring that processes, systems, and products meet predefined quality standards and regulatory requirements.
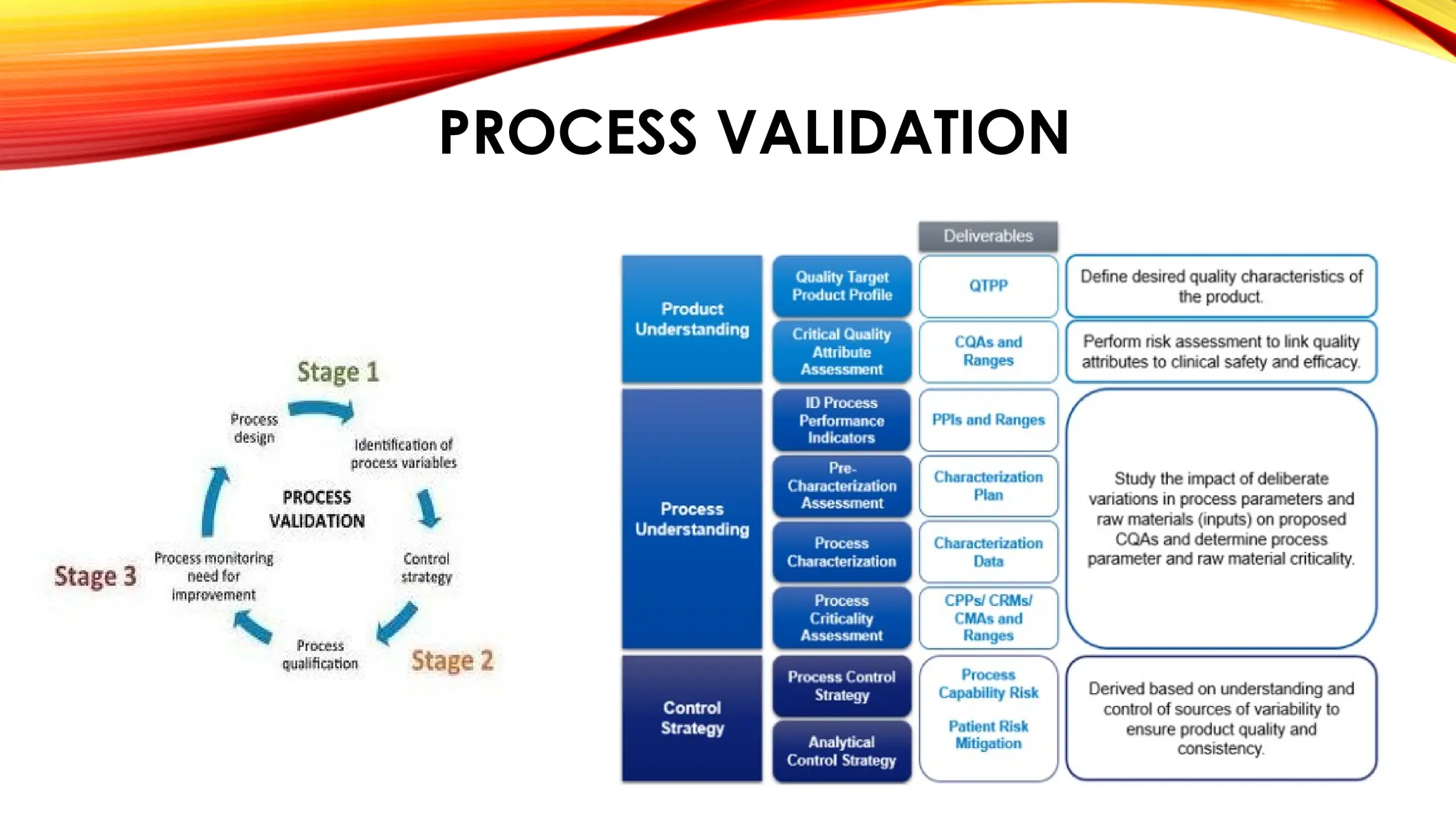
Overview On Validation In Pharmaceuticals Pptx Finalization of the validation process: a validation report must be prepared at the conclusion of validation activities. it should summarize and reference all protocols and results. obviously, a conclusion is required: “is the process valid”, but strangely often omitted. Validation in the pharmaceutical industry is a systematic approach to ensuring that processes, systems, and products meet predefined quality standards and regulatory requirements. The document discusses pharmaceutical validation, including definitions, types, and elements of validation. it provides definitions of validation from who, fda, and ich. Validation in the pharmaceutical and medical device industry is crucial for demonstrating consistent results. this process ensures compliance with regulations and good manufacturing practices for procedures, processes, and equipment. learn about the importance, history, and elements of validation. Explore our comprehensive validation process presentation that is fully editable and customizable. enhance your understanding and effectively communicate key concepts with ease. “validation is a process of demonstrating, through documented evidence, that a process, procedure, method, piece of equipment, or facility will consistently produce a product or result that meets predetermined specifications and quality attributes.”.

Overview On Validation In Pharmaceuticals Pptx The document discusses pharmaceutical validation, including definitions, types, and elements of validation. it provides definitions of validation from who, fda, and ich. Validation in the pharmaceutical and medical device industry is crucial for demonstrating consistent results. this process ensures compliance with regulations and good manufacturing practices for procedures, processes, and equipment. learn about the importance, history, and elements of validation. Explore our comprehensive validation process presentation that is fully editable and customizable. enhance your understanding and effectively communicate key concepts with ease. “validation is a process of demonstrating, through documented evidence, that a process, procedure, method, piece of equipment, or facility will consistently produce a product or result that meets predetermined specifications and quality attributes.”.
Overview On Validation In Pharmaceuticals Pptx Explore our comprehensive validation process presentation that is fully editable and customizable. enhance your understanding and effectively communicate key concepts with ease. “validation is a process of demonstrating, through documented evidence, that a process, procedure, method, piece of equipment, or facility will consistently produce a product or result that meets predetermined specifications and quality attributes.”.
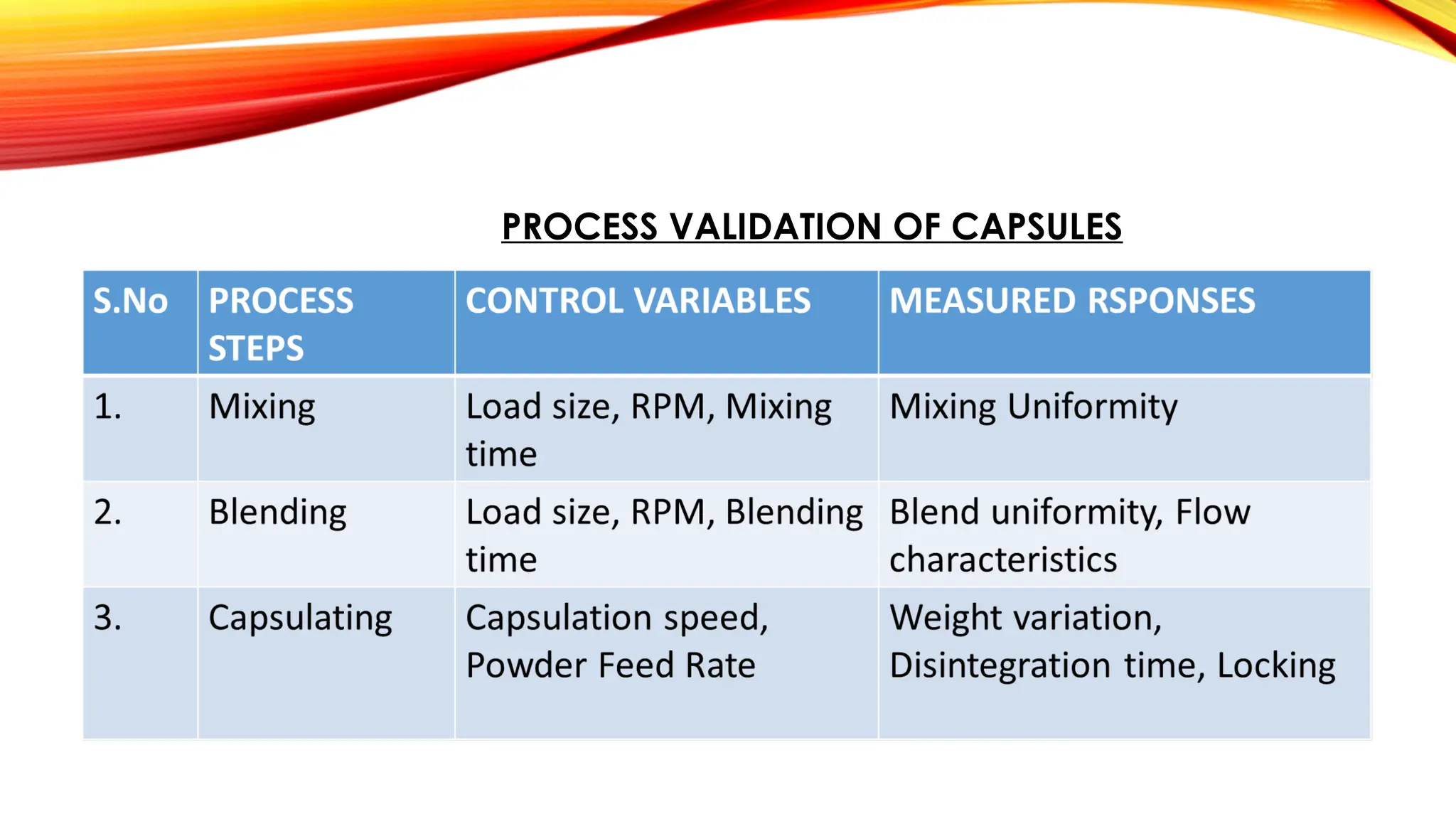
Overview On Validation In Pharmaceuticals Pptx
Comments are closed.