Pharma Focus Phase2

Pharma Focus Phase2 Innovation in pharma means designing within a framework that allows creativity to thrive. at phase2, we help you move faster, engage deeper, and confidently bring your digital vision to life, knowing compliance is built into every step. Reporting on the pharma industry, drug development & clinical trials, manufacturing, logistics, market access, and patients. from breaking stories to interviews with senior executives, we look at regulations & trends to provide useful market intelligence.

Pharma Focus Phase2 Nxera pharma has achieved a second development milestone under its multitarget collaboration and licence agreement with eli lilly focused on diabetes and other metabolic diseases. Abbvie (nyse: abbv) today announced that late breaking results from the phase 2 imgn853 0420 trial will be presented in an oral session at the 2026 society of gynecologic oncology (sgo) annual. Ascendis pharma (nasdaqgs:asnd) is back in focus after new coach phase 2 data on its transcon combination therapy in achondroplasia and the launch of yuviwel in the us following fda orphan drug. Centessa is positioning orx750 as a “best‑in‑class” orexin‑2 agonist focused on efficacy and duration of action, plans a phase 2 update this quarter and aims to start data‑driven registration studies “this quarter,” while retaining the option to continue dose escalation and add higher doses into registrational plans.

Pharma Focus Phase2 Ascendis pharma (nasdaqgs:asnd) is back in focus after new coach phase 2 data on its transcon combination therapy in achondroplasia and the launch of yuviwel in the us following fda orphan drug. Centessa is positioning orx750 as a “best‑in‑class” orexin‑2 agonist focused on efficacy and duration of action, plans a phase 2 update this quarter and aims to start data‑driven registration studies “this quarter,” while retaining the option to continue dose escalation and add higher doses into registrational plans. Christopher cargill, president and ceo of nxera pharma, commented: “the initiation of this phase 2 study with nbi ‘570, the second nxwave™ designed compound in neurocrine’s muscarinic agonist portfolio to advance into phase 2 clinical trials, represents another important step forward in our highly productive collaboration. Tarsus pharmaceuticals has already dosed the first participant in its phase 2 calliope trial of tp 05, an oral lotilaner based therapy being tested in about 700 at risk adults across u.s. lyme endemic regions, where no fda approved drug prophylaxis currently exists. this move extends tarsus beyond eye care into infectious disease prevention, targeting a large u.s. population considered at. Stay updated with the latest trends in pharma, including r&d, clinical trials, and drug manufacturing. explore expert insights and innovations in every issue. Recce pharmaceuticals has announced positive patient data from its phase 2 clinical trial into recce 327 (r327), for the treatment of acute bacterial skin and skin structure infections (absssis), including diabetic foot infections (dfi).
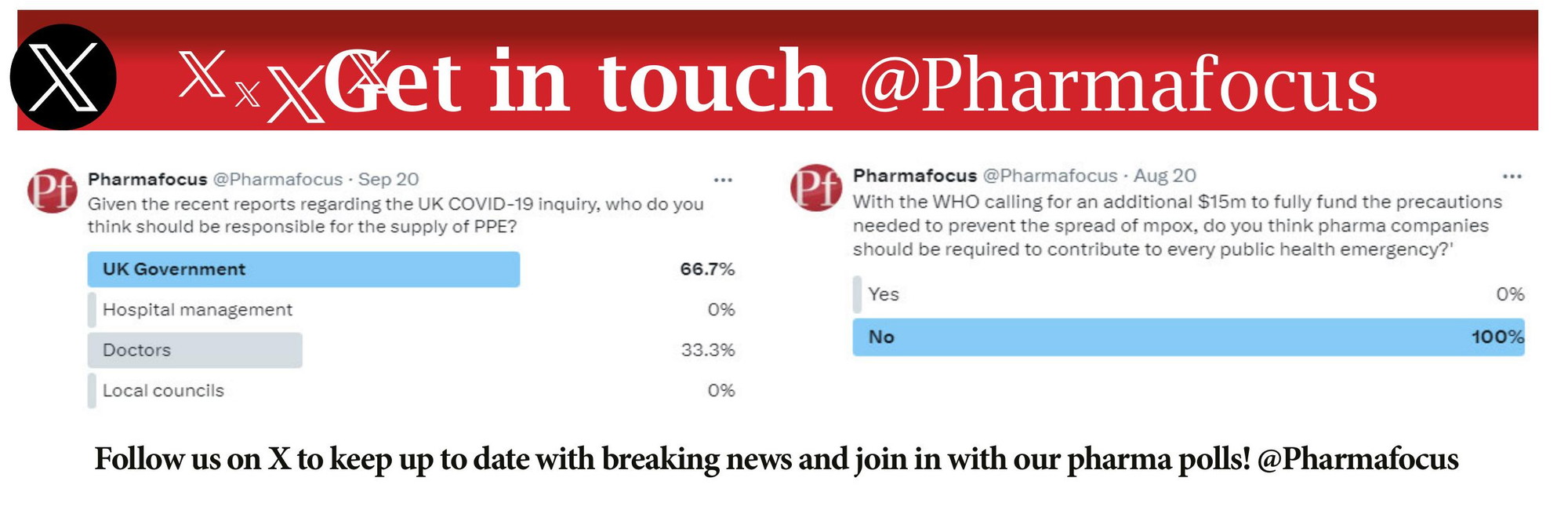
Pharma Focus Christopher cargill, president and ceo of nxera pharma, commented: “the initiation of this phase 2 study with nbi ‘570, the second nxwave™ designed compound in neurocrine’s muscarinic agonist portfolio to advance into phase 2 clinical trials, represents another important step forward in our highly productive collaboration. Tarsus pharmaceuticals has already dosed the first participant in its phase 2 calliope trial of tp 05, an oral lotilaner based therapy being tested in about 700 at risk adults across u.s. lyme endemic regions, where no fda approved drug prophylaxis currently exists. this move extends tarsus beyond eye care into infectious disease prevention, targeting a large u.s. population considered at. Stay updated with the latest trends in pharma, including r&d, clinical trials, and drug manufacturing. explore expert insights and innovations in every issue. Recce pharmaceuticals has announced positive patient data from its phase 2 clinical trial into recce 327 (r327), for the treatment of acute bacterial skin and skin structure infections (absssis), including diabetic foot infections (dfi).

Pharma Focus Stay updated with the latest trends in pharma, including r&d, clinical trials, and drug manufacturing. explore expert insights and innovations in every issue. Recce pharmaceuticals has announced positive patient data from its phase 2 clinical trial into recce 327 (r327), for the treatment of acute bacterial skin and skin structure infections (absssis), including diabetic foot infections (dfi).
Comments are closed.